- Home
- Industry
- Applications and case studies
- Engineering
Engineering
Innovative materials, such as composites and alloys that can withstand ageing and extreme conditions, are driving advances in the aerospace, nuclear and numerous other industries. Understanding the behaviour of materials during their formation and use is crucial to allow manufacturers to develop new materials with tailored properties, such as those with reduced environmental impact. Non-destructive synchrotron X-ray techniques offered by the ESRF allow industry scientists to correlate a material’s micro- and nanostructure with its properties.
Complementary to static structure determination, the ESRF allows the study of materials during their synthesis or under realistic working conditions, which helps manufacturers identify and exploit a material’s properties in new ways.
- Track changes occurring inside materials during formation on the scale of milliseconds.
- Strain and stress analysis of materials using energy dispersive X-ray diffraction.
- Conducting damage and failure testing on coatings using X-ray microtomography.
CASE STUDIES
Company
TU Delft together with steel manufacturers
Challenge
The Twin Towers collapsed in 2001 because the steel that kept them standing couldn´t resist the fire. The changes in temperature modified the microstructure of the steel and it lost its strength, making the towers crumble. Knowing what happens inside steel (the nucleation process) when it is submitted to high temperatures could help find a better composition for this alloy.
Sample
Steel.
Solution
3D X-ray diffraction on ID11 has proven a unique technique to track the nucleation process inside steel as it is submitted to temperatures of 1000 C. Scientists came up with a furnace that could be compatible with the beamline and that had temperature control. They found that nucleation, which is a key process in metals, happens quicker than literature had predicted: there are special places, like grain corners or edges in the material where nucleation takes place more easily than other areas. Amongst these corners and edges there are ones that are preferred for nucleation. Controlling the nucleation means controlling the properties of the material.
Also, the crystals in the steel become coarser at higher temperatures, which decreases the material's strength. Scientists are currently testing the replacement of potentially scarce elements in the alloying process of steel by other elements more widely available.
Benefits
A better “recipe” for steel using less alloying elements would make the material more fire-resistant, less expensive and easier to recycle whilst maintaining its properties.
Sharma H., et al, Scientific Reports, DOI: 10.1038/srep30860

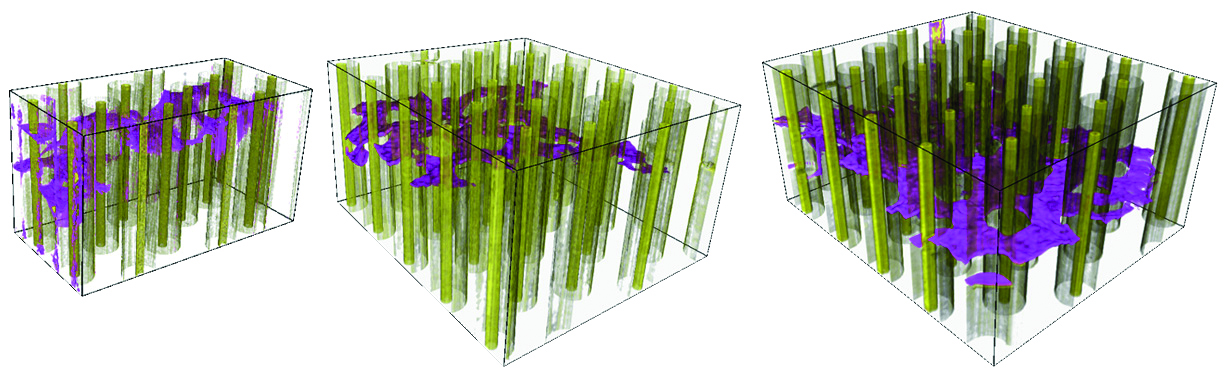 |
|
3D crack-tip microscopy shows a crack (purple) growing in a composite material containing silicon carbide fibres. |
Company
University of Mancherster and Rolls-Royce
Challenge
Crack propagation in metallic materials is well understood. But aircraft manufacturers are increasingly turning to more complicated composite materials that are lighter, stronger and can operate at higher temperatures. Lower weight reduces fuel consumption, while higher engine operating temperatures allow aeroengines to be more efficient. The challenge is to understand how cracks propagate in such materials.
Sample
Titanium reinforced with silicon carbide fibres. This composite material can operate at higher temperatures than titanium alone, making it a promising candidate for jet engine parts.
Solution
Synchrotron X-rays penetrate tens of millimetres into a sample where the behaviour of cracks can be very different - this in contrast to eectron microscopy which only reveals the surface features of micro-cracks. On beamline ID15, scientists can use imaging, to see how cracks grow, and diffraction, which tells them about the local stresses that the cracks grow under.
Benefits
The ability to monitor cracks under load at high temperatures allows researchers to evaluate the potential of these materials under realistic conditions. It also helps to make realistic estimates
of the lifetime of existing components and to design safer, more crack-resistant materials for the future.
Better knowledge of crack propagation transfers directly to other industries in which failure is unacceptable, notably the nuclear industry.
Company
ECOCEM, LMT (ENS Cachan, CNRS, Université Paris Saclay), LMDC (Université de Toulouse INSA/UPS Génie Civil).
ECOCEM Materials is the first independent producer of Ground Granulated Blast Furnace Slag (GGBS) in Europe. ECOCEM recycles a by-product from steel industry: granulated slag from a blast furnace which is in glassy form. The aim is to produce concrete with lower environmental impact: 1 ton of ground slag in the concrete industry avoids the emission of 750 kg of CO2 due to the production of Portland cement.
Challenge
In the quest for environmentally friendly materials, ECOCEM is using slag rich concrete, mortar and other materials. These materials might display a blue/green colour when they get consolidated. When the concrete is exposed to air, the blue/green colour turns to grey (in the case of cement concrete) or white (slag concrete). However, if the concrete is let to set for a long time in a form work or cured under water, it keeps the colour for up to its entire service life. This specific colour is concerning in applications where visual aesthetics are important. The origin of this blue/green tinge was not resolved until now, even if it has been attributed to the presence of polysulphur species in the slag, but without any scientific evidence.
Sample
- Ground granulated blast furnace slag (GGBS). It is an alumino-silicate glass containing in particular less than 1% of total sulphur species.
- Non-ground granulated slag collected from a steel factory without grinding.
- The last sample of slag was ground under nitrogen atmosphere in the laboratory to avoid sulphur oxidation.
 |
|
A cafe built with ECOCEM cement. Credits: ECOCEM. |
Solution
In order to find out the origin of this coloration, scientists went to ID21 and used K-edge X-ray absorption near edge structure (XANES) spectroscopy to follow the evolution of the speciation of sulphur as the slag was hydrated.
The team used three methods to activate the slag: thermal, Portland cement and sodium silicate. First, they compared each method and then they compared the XANES results on blue and white samples and on zones that were not coloured.
They found that, independently of the activating method, the blue colour was related to the presence of a pre-peak in the corresponding XANES spectrum. This did not happen anywhere else in the samples. They linked this pre-peak to the presence of the polysulphur radical anion S3- (as shown in the figure). This blue chromophore is known to be in particular at the origin of the deep blue of the stone lapis lazuli or the ultramarine pigment (derived from the Lazurite mineral).
As for each specific sample, the team found that the oxidation state of sulphur in a granulated slag obtained just after quenching was higher than in a ground slag or when ground under nitrogen atmosphere. This suggests that the superficial sulphur is oxidized under quenching.
Benefits
This research did not exclude the possibility of other elements influencing the coloration of slag-based concretes, in particular the presence of transition metals, but it showed clearly that the trisulphur radical anion is present and may play a major role.
In-depth understanding of the coloration mechanism was the key point if an effective decolorizing treatment was to be found. Through the study in ESRF by XANES, ECOCEM finally found an explanation of the blue/green colour: the presence of S3- in the concrete matrix. This study helped ECOCEM to explain clearly the reason of blue colour formation to their clients. And it also helped indirectly ECOCEM to find the solutions to eliminate the blue/green colour.
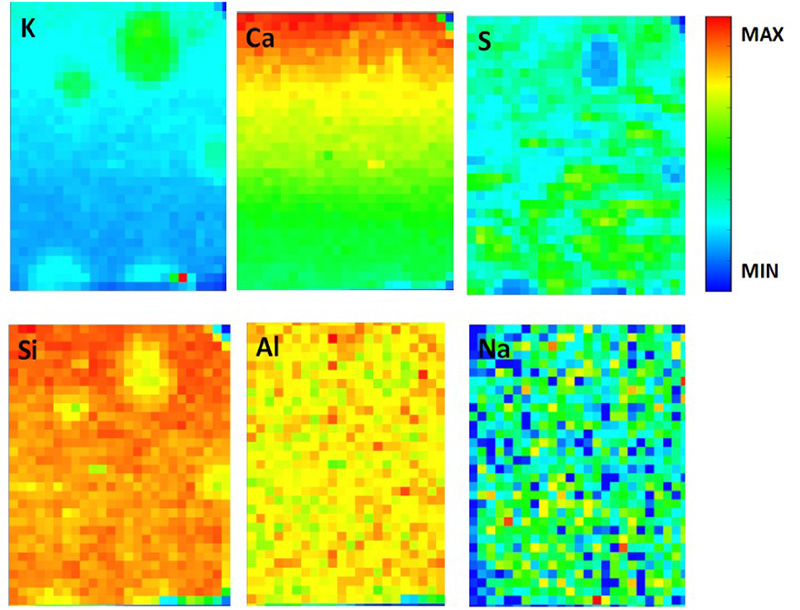 |
|
Elemental distribution obtained by μ-XRF with an exciting beam of 4040 eV for K and Ca, and 2480 eV for S, Si, Al, O and Na obtained by μ-XRF with an exciting beam, map size 2.6 x 3.5 mm2 with a step of 100 µm. |
Reference:
Chaouche M, Gao XX, Cry M, Cotte M and Frouin L, On the origin of the blue/green color of blast-furnace slag-based materials: Sulfur K-edge XANES investigation, J Am Ceram Soc. 2017; 1-10. doi: 10.1111/jace.14670.



