- Home
- News
- Spotlight on Science
- Structure of membrane...
Structure of membrane lipid modifying protein provides starting point for development of drugs to fight RNA viruses and malaria
18-06-2014
The crystal structure of the tertiary complex of a phosphatidylinositol 4-kinase with a small guanosine triphosphatase and its effector provides insight into processes that require the remodelling and expansion of phosphatidylinositol 4-phosphate containing membranes such as the intracellular development of malarial pathogens and the replication of a wide range of RNA viruses. The structure of the tertiary complex obtained provides a good starting point for the development of inhibitors to fight RNA virus replication and malaria.
Compartmentalisation of any eukaryotic cell is crucial for all cellular processes and compartment identity is usually given by specific lipids -such as phosphoinositides- located in the membrane. Phosphorylation of phosphatidylinositol results in phosphatidylinositol 4-phosphate (PI4P), a reaction performed by a family of enzymes called phosphatidylinositol 4-kinases (PI4Ks). PI4KIIIβ is a prominent mammalian member of this family and localises primarily at the Golgi apparatus, the organelle responsible for the modifying, sorting and packing of proteins for cell secretion or for use within the cell. PI4KIIIβ, essential for Golgi formation and function, not only acts as an enzyme to produce PI4P but also interacts non-catalytically with other proteins such as Rab11, a small GTPase that performs various functions in membrane trafficking by binding specific effector molecules.
PI4KIIIβ is of high medical relevance: the replication of several RNA viruses including hepatitis C, SARS corona or enteroviruses is accomplished by the hijacking of PI4KIIIβ activity [1, 2] and compounds that target PI4KIIIβ have been shown to inhibit the replication of such pathogens; molecules that inhibit P. falciparum PI4KIIIβ activity have been shown to be effective agents against malaria [3]. Here, inhibiting PI4KIIIβ prevents membrane ingression during a particular phase of the parasite's life cycle. Unfortunately though, point mutants of both PI4KIIIβ and Rab11 were resistant to such inhibitors.
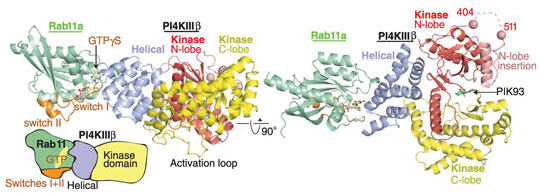
To provide insight as to how PI4KIIIβ interacts with Rab11 and enables communication of the latter with effector molecules, the crystal structure of a modified form PI4KIIIβ in complex with GTP-bound Rab11a was determined to a resolution of 2.9 Å using data obtained from beamlines ID23-2 and ID29 (Figure 1). As well as revealing that the structure of PI4KIIIβ consists of a helical solenoid domain linked to a much larger kinase domain made up of two lobes, the crystal structure obtained also revealed that the regions required for the binding of a Rab11a effector remain available in the complex. This suggests PI4KIIIβ could bind simultaneously both to Rab11a and its effector molecules.
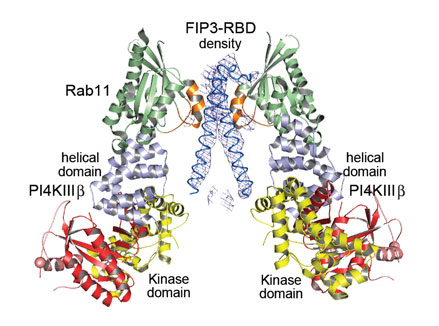
The crystal structure of the ternary complex between PI4KIIIβ, Rab11a and the Rab-binding domain (RBD) of the effector molecule FIP3, obtained to a resolution of 6 Å (Figure 2), confirms this hypothesis and shows that in the ternary complex the FIP3 effector specifically interacts with the switch regions of Rab11a. Taken together, the two crystal structures described here provide an excellent model for recruitment of Rab11 and its effectors to PI4P-enriched membranes by PI4KIIIβ. They also provide significant clues as to how to disrupt interactions that are crucial to this process and thus pave the way for the design of molecules that may act as powerful drugs against malaria and enteroviral infections.
Principal publication and authors
Structures of PI4KIIIβ complexes show simultaneous recruitment of Rab11 and its effectors, J.E. Burke (a), A.J. Inglis (a), O. Perisic (a), G.R. Masson (a), S.H. McLaughlin (a), F. Rutagarina (b), K.M. Shokat (b), R.L. Williams (a), Science 344, 1035-1038 (2014).
(a) Medical Research Council (MRC) Laboratory of Molecular Biology, Cambridge (UK)
(b) Howard Hughes Medical Institute and Department of Cellular and Molecular Pharmacology, University of California, San Francisco (USA)
References
[1] N.-Y. Hsu et al., Cell 141, 799-811 (2010).
[2] N. Altan-Bonnet, T. Balla, Trends Biochem. Sci. 37, 293-302 (2012).
[3] C.W. McNamara et al., Nature 504, 248-253 (2013).
Top image: Crystal structure of the ternary complex between PI4KIIIβ, Rab11a and FIP3-RBD (Rab-binding domain).