- Home
- News
- Spotlight on Science
- Revealing the nature...
Revealing the nature of nanostructured reagents in organic synthesis
09-10-2018
Nanostructured nickel thiolates were used as the unique reagents for the synthesis of organic sulphides in C-S cross-coupling. Structural studies at a synchrotron light source and multi-level monitoring of the reaction revealed the key reactivity features of the thiolate particles.
Inorganic and hybrid nanostructured materials based on transition metals have made an outstanding contribution to the fields of materials science, catalysis, energy research and others. Due to the complex multi-level structure of nanomaterials, the need for their structural characterisation gave rise to the development of a number of advanced techniques including electron microscopy and X-ray spectroscopy methods. In the field of nano-research, particular attention is paid to the fundamental understanding of the function of nanostructured materials in chemical transformations at different levels: atomic, molecular, nano- and micrometre. A comprehensive study of the structural features determining the reactivity of compounds with multi-level organisation is urgently needed for the construction of materials with desired properties. For example, nanoparticle-mediated catalytic transformations in solutions represented a black box of chemistry for many years due to a large amount of convergent and divergent routes of catalyst transformation in the course of the reaction [1-2].
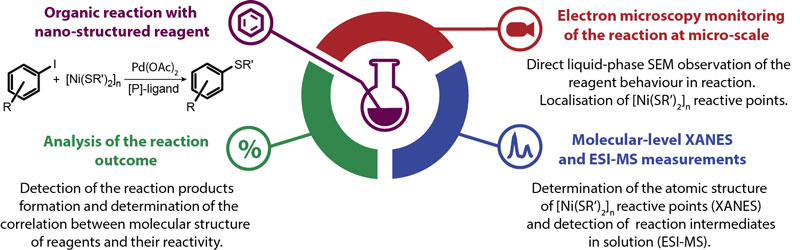
In this work, a complex approach for the in situ characterisation of nanostructured materials reactivity was introduced. The aim of this research was to employ nanostructured metal-organic particles as reagents for a widely-used chemical transformation and to reveal their mechanism of action in the reaction mixture. Nickel thiolate coordination polymers based on nickel central metal atoms and sulphur-containing organic groups with chemical formula [Ni(SR)2]n (where R – organic substituent) also known as “nano-salts” [3] were used as a source of reactive groups in the carbon-sulphur bond formation reaction in solution leading to organic sulphides. Starting from the bulk process in a flask (where the influence of the nature of substituents on the reaction rate and product yield was determined via conventional reaction monitoring) the next step was to obtain a deeper understanding of the behaviour of the reagent particles in the chemical transformation (Figure 1).
 |
|
Figure 1. Illustration of the complex approach for the study of nanostructured reagents in organic carbon-sulphur bond formation reaction at different stages of the reaction. |
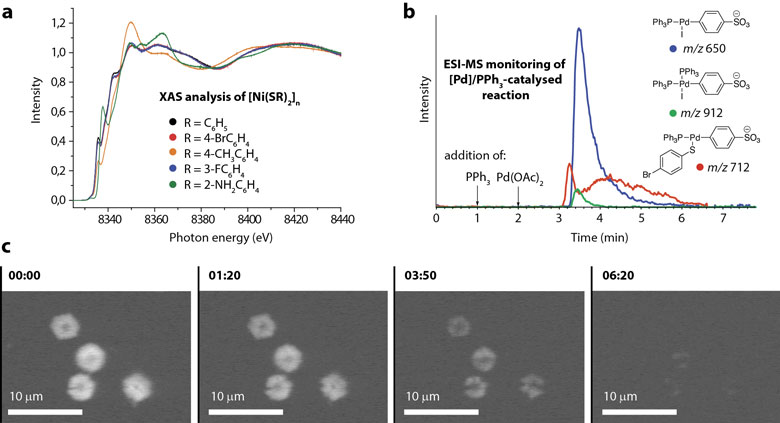
High-energy-resolution fluorescence-detected X-ray absorption spectroscopy (HERFD-XAS) at the nickel K-edge was used at beamline ID26 to elucidate the chemical state and coordination environment of metal atoms in the particles of nanostructured reagent. Generally, Ni2+ centres were surrounded by four sulphur atoms forming the coordination polymer unit in square planar geometry (Figure 2a). In some cases, interactions with sulphur and other coordinating atoms (e.g. nitrogen) were also detected. Interestingly, the electronic structure and the local atomic environment of the central Ni atoms were almost identical in samples of nickel-containing coordination polymers despite their completely different morphologies. Similar spectral patterns were observed for the simple [Ni(SC6H5)2]n compound and its fluorine- and bromine-containing analogues. All compounds studied featured planar subunits. This observation led to the suggestion that the nickel thiolate particles had a hierarchical structure and regions with specific reactivity.
Dynamic electron microscopy experiments, which were carried out directly on the liquid phase, detected the non-uniformity of reagent particle dissolution during the reaction. This result indicated the existence of particular reactive regions at the interface between particles and solution which determined the micro- and nano-scale effects of the reagent structure on the reaction outcome (Figure 2c). Moving deeper to the molecular level, electrospray ionisation mass spectrometry (ESI-MS) was employed to detect reactive species in solution. On-line monitoring allowed key intermediates of the transformation to be detected in real time and their formation and consumption traced. On the basis of the observed kinetics, which was non-typical for a classical reaction in solution, the hypothesis of preferential reactivity of the interface between particle surface and solution could be verified (Figure 2b).
To summarise, a novel type of reagent with nano-scale properties was employed for a highly-demanded process in organic synthesis. The mechanism of the chemical transformation was unravelled using a step-by-step approach starting from monitoring the bulk reaction and ending with capturing molecular-level features of the reaction mixture.
Principal publication and authors
Exploring the performance of nanostructured reagents with organic-group-defined morphology in cross-coupling reaction, A.S. Kashin, E.S. Degtyareva, D.B. Eremin, V.P. Ananikov, Nat. Commun. 9, 2936 (2018); doi: 10.1038/s41467-018-05350-x.
Zelinsky Institute of Organic Chemistry, Moscow (Russia)
References
[1] D.B. Eremin et al., Coord. Chem. Rev. 346, 2 (2017).
[2] A.S. Kashin et al., J. Org. Chem. 78, 11117 (2013).
[3] V.P. Ananikov et al., Dalton Trans. 40, 4011 (2011).