- Home
- News
- Spotlight on Science
- 3D multiscale imaging...
3D multiscale imaging of human vocal folds
11-12-2018
The 3D architecture of human vocal folds was revealed ex vivo using synchrotron X-ray microtomography. This provides a better understanding of vocal-fold micromechanics and their outstanding vibratory characteristics.
Although only a few millimetres in size, human vocal folds have remarkable vibro-mechanical properties allowing them to generate an outstanding range of sounds compared to that of a musical instrument. Two properties of vocal-fold tissues make their sound characteristics possible [1]:
- the ability to endure large, reversible deformations during phonation, typically stretching between 10 and 50% during a glide or intonational variation. Such an elastic recoil takes place despite the periodic collisions and mechanical stresses to which the vocal folds are subjected for many hours daily.
- the ability to vibrate with a fundamental frequency ranging from less than 50 Hz to more than 1500 Hz (mean value of 125 Hz/210 Hz in the speech of men/women), related to the tonal pitch. This frequency is much higher than that of other biological oscillators (heart/respiratory frequency at/below 1 Hz).
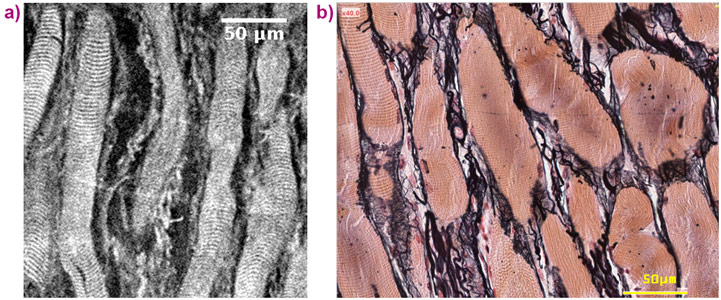
These outstanding properties are mainly inherited from the complex and multiscale structure of vocal-fold tissues, which are very difficult to investigate experimentally. In particular, the 3D observation of excised vocal folds at the (sub-)micron scale still presents challenges using either confocal microscopy (limited depth of field), magnetic resonance imaging (limited spatial resolution) or X-ray absorption microtomography in absorption mode (limited phase contrast) [2,3].
High-resolution synchrotron X-ray microtomography with phase retrieval (voxel sizes between 0.65 and 13 μm) at beamline ID19 has now been used to obtain the first ex vivo 3D images of human vocal-fold tissues at multiple length scales (see Figure 1). Various structure descriptors were extracted from the images: geometry of the vocal folds at rest or in a stretched phonatory-like position; shape and size of their layered fibrous architectures within the lamina propria (upper vocal-fold loose connective tissue) and the vocalis muscle; orientation, shape and size of muscle fibres as well as collagen and elastin fibre bundles constituting these layers (see Figure 2). In particular, it was shown that the intermediate lamina propria sublayer, marked by a dense network of straighter collagen-elastin fibres, is confined to a region likely to endure the highest collision stresses during phonation. Observations support the hypothesis that regions exposed to higher stresses in the vocal tissues are characterised by a higher density of fibrous proteins produced to strengthen the extracellular matrix. In addition, for the first time, the 3D structural anisotropy of fibrous networks in the lamina propria and in the vocalis could be quantified using 3D second-order fibre orientation tensors. Although characterised only in 2D so far, the degree and magnitude of such anisotropic properties are known to strongly impact the predictions of the vocal-fold fundamental frequency of vibration [2], as well as the stress responses of the tissue under compression modes, which is critical to better understand vocal-fold collision.
The methodology used in this study yields a quantified description of the complex anatomy and histology of the vocal folds, and allows an evaluation of recent micro-mechanical models of voice biomechanics [2-4]. Finally, with a scan duration of 1-2 minutes, the experimental procedure provides promising insights to further track the deformation and rearrangement of muscle and collagen/elastin fibres during mechanical load at the (sub-)micron scale. Such information is crucial to gain an in-depth understanding of the link between the micro-mechanics of vocal-fold tissues and their unique vibratory properties, and to take the next step towards the development of new biomimetic oscillators.
Principal publication and authors
3D multiscale imaging of human vocal folds using synchrotron X-ray microtomography in phase retrieval mode, L. Bailly (a), T. Cochereau (a,b), L. Orgéas (a), N. Henrich Bernardoni (b), S. Rolland du Roscoat (a), A. McLeer-Florin (c), Y. Robert (d), X. Laval (b), T. Laurencin (a), P. Chaffanjon (d,b), B. Fayard (e), E. Boller (f), Scientific Reports 8, 14003 (2018); doi: 10.1038/s41598-018-31849-w.
(a) Univ. Grenoble Alpes, CNRS, Grenoble INP, 3SR, Grenoble (France)
(b) Univ. Grenoble Alpes, CNRS, Grenoble INP, GIPSA-lab, Grenoble (France)
(c) Univ. Grenoble Alpes, CHU Grenoble Alpes, Histology Lab, IAB, Grenoble (France)
(d) Univ. Grenoble Alpes, CHU Grenoble Alpes, LADAF, Grenoble (France)
(e) Novitom, Grenoble (France)
(f) ESRF
References
[1] I.R. Titze, Principles of voice production, National Center for Voice and Speech (2000).
[2] J.E. Kelleher et al., The Journal of the Acoustical Society of America 133, 1625–36 (2013).
[3] A.K. Miri et al., The Laryngoscope 122, 356–63 (2012).
[4] A.K. Miri et al., Acta Biomaterialia 9, 7957–7967 (2013).