- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- High Resolution and Resonance Scattering
- Effect of Pt and Pd Nanoparticles on the Gas Sensitivity of Polycrystalline SnO2 Thin Films: In situ XAS Study
Effect of Pt and Pd Nanoparticles on the Gas Sensitivity of Polycrystalline SnO2 Thin Films: In situ XAS Study
Polycrystalline SnO2 films are the materials of choice for the development of resistive gas sensors, which are used for the detection of toxic and explosive gases (CO, H2, NOx etc) [1]. The sensor properties are based on the fact that the electrical conductivity of SnO2 films depends on the atmosphere composition. SnO2 is an n-type wide band gap semiconductor. Adsorption of oxygen species creates trapped charges on the SnO2 surface. Adsorption and consecutive redox reaction of a target gas with the adsorbed oxygen species leads to modulation of the quantity of charge and a variation of conductivity. Addition of noble metals modifies the electronic and catalytic properties of the SnO2 by forming nanoparticles on the oxide surface. This process greatly enhances the sensitivity and decreases the working temperature.
In spite of wide practical applications, the mechanism of resistive gas sensors is still under discussion. Most of the studies on the reactivity of sensor materials were done ex situ. This is not favourable since the chemical state of the sensor surface is extremely sensitive toward the atmosphere composition even at room temperature [1]. For SnO2 films containing Pt nanoparticles it was previously observed using XANES spectroscopy that the Pt oxidation state strongly depends on the size of the nanoparticles and the presence of CO in the atmosphere[2]. In the present work the effect of Pt and Pd on the mechanism of SnO2 gas sensitivity toward H2 was studied at ID26 by XAS in operando conditions.
The SnO2 porous films (thickness 0.8-1.0 µm) were synthesised by aerosol pyrolysis [2]. Four systems were studied: pure SnO2film (referred to as Sn1 in the following) and the SnO2 films containing 2.0 at% Pt (Pt1), 6.3 at% Pt (Pt2) and 1.2 at% Pd (Pd1). The fluorescence XAS at Pt L3- and Pd K-edges were acquired using quick EXAFS (30 s per spectrum) simultaneously with the conductivity measurements. The films were exposed to 20% O2 in He (O2), 1000ppm H2 in He (H2), and 1000ppm H2 + 20% O2 in He (H2 + O2).
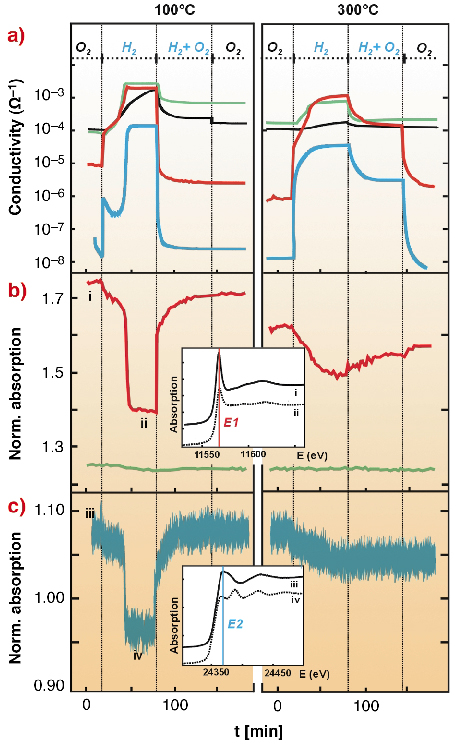
We observed different mechanisms of gas sensitivity at high temperature (200°-300°C) and at low temperature (50°-100°C). We found that the Pt1 and Pd1 systems are much more sensitive to the presence of H2 than Sn1 and Pt2 (Figure 6). The variation of normalised absorption at fixed energies near the Pt L3- and Pd K-edges (Figures 6b and 6c) indicates the change of platinum and palladium oxidation states. Comparing experimental data with the spectra of reference compounds (metals and oxides) one can conclude that conductivity variations of Pt1 and Pd1 are accompanied by synchronous oxidation and reduction of platinum and palladium. Under the same conditions, platinum in Pt2 is almost metallic and non-reactive. This correlates with the fact that the Pt2 film contains bigger (2.3 nm) Pt nanoparticles than Pt 1 (1.7nm) [2].
 |
|
Fig. 6: Conductivity (a) and normalised absorption at E1 = 11568.5 eV (b) and E2 = 24368.6 eV (c) for Sn1 (black curve), Pt1 (red), Pt2 (green) and Pd1 (blue) in different atmospheres. The insets show the XANES region for Pt and Pd. |
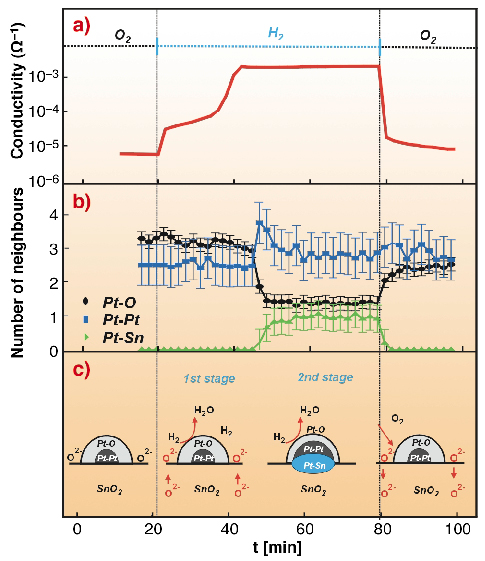
The kinetics of the conductivity variation of Pt1 and Pd1 films in H2 give a similar picture. Two stages of conductivity increase are observed at 300°C and only the second one is related to a variation of Pt and Pd oxidation state. For the Pt1 film, evaluation of the EXAFS data indicates not only the reduction of platinum but also of the SnO2 matrix (Pt-Sn bonds in Figure 7). At low temperatures the conductivity of the Pt1 and Pd1 systems increases much faster than at high temperatures but the state of platinum and palladium varies very little. High sensitivity of Pt1 and Pd1 films towards H2 + O2 was also observed at 50-100°C.
 |
|
Fig. 7: (a) Conductivity; (b) the number of neighbours in the first shell of platinum calculated from EXAFS and (c) the proposed model for the Pt1 system at 300°C. |
We propose that the difference between the high temperature and the low temperature mechanisms is related to the oxygen mobility in the SnO2 matrix. At low temperatures the redox reactions take place only at the SnO2 surface that leads to the fast variation of the quantity of charge trapped at the SnO2 surface by acceptor states including the states produced by Pt and Pd nanoparticles [1]. At high temperatures the participation of the bulk oxygen slows down the modulation of this charge.
References
[1] L. Moris, D.E. Williams, J. Phys. Chem. B, 105, 7272-7279, (2001).
[2] M. Gaidi, J.L. Hazemann, I. Matko, B. Chenevier, M. Rumyantseva, A. Gaskov and M. Labeau, J. Electochem. Soc., 147, 3131-3138, (2000).
Authors
O. Safonova (a), T. Neisius (a), B. Chenevier (b), A. Gaskov (c), M. Labeau (b).
(a) ESRF
(b) LMGP, Institut National Polytechnique de Grenoble (France).
(c) Chemistry Department, Moscow State University (Russia).



