- Home
- News
- Spotlight on Science
- A promiscuous intermediate...
A promiscuous intermediate underlies the evolution of single copy transcription factors
04-03-2014
As organisms evolve and branch out into new species, the complexity of the underlying gene regulation network expands. A multidisciplinary team used cutting edge techniques, including X-ray crystallography at beamline ID23-EH2 to elucidate a mechanism for this expansion.
Share
A class of proteins known as transcription factors (TFs) is central to the evolution of organisms. TFs exert their control on the cell by binding specifically to DNA sequences and causing a cascade of downstream events to occur.
A key question in the evolution of TFs is how they can evolve new DNA specificities. This is complicated by the fact that altering the target DNA sequence or the specificity of the transcription factor will in most cases change the binding properties of the TF towards its original target DNA sequence, which will in turn prevent the normal downstream events from occurring. A simple solution to this problem is the duplication of genes, followed by the evolution of new properties (most notably DNA binding specificity) of one copy of the gene. In this scenario, the organism will continue to survive because it has a working copy of both the DNA and TF, which frees the second copy of the TF to evolve new binding specificities. However, in cases where only a single copy of the gene exists in the cell, it is less obvious how changes can occur without deleterious effects. In this work, LEAFY, a generally single copy master regulator of floral development, was used as a model system. A key discovery which enabled this research was the identification of ancient forms of LEAFY from Hornworts and algae. The DNA binding preferences of these proteins and other previously described forms from angiosperms and gymnosperms were determined using a technique called SELEX (systematic evolution of ligands by exponential enrichment). From the data, in combination with classical biochemical binding assays, a “family tree” of DNA binding specificity could be assembled. The family tree showed that there are at least three different types of DNA recognition sites: type I, II and III.
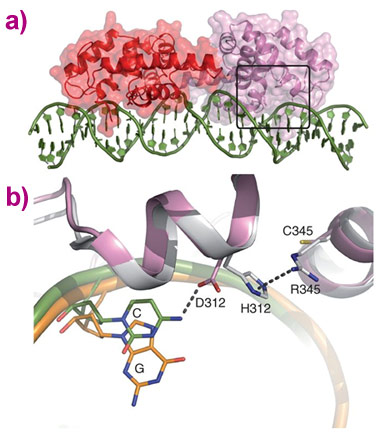
Using the microfocus molecular crystallography beamline ID23-2 [1] and a special data collection method to reduce radiation damage, the crystal structure of one of these family members (from moss) was determined, in complex with its preferred (type II) DNA sequence. When compared to an earlier structure of LEAFY in complex with a type I sequence [2], this structure revealed that an additional contact is made between the protein and DNA, and provided a molecular explanation for the altered DNA specificity compared to other LEAFY forms (Figure 1).
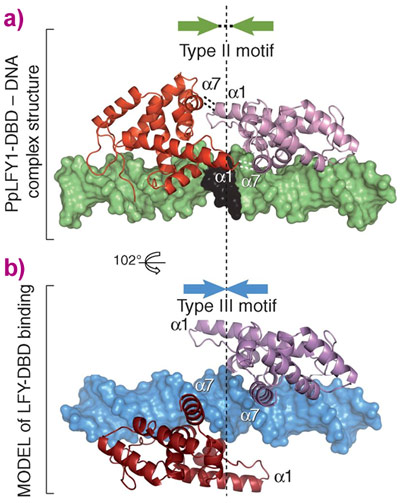
Equally important was the elucidation of the molecular determinants of primitive LEAFY binding to the type III motifs. Type III motifs differ dramatically from Type I/II in that a central region in the recognised DNA is absent. Using SELEX, biochemical binding assays and the Type I and Type II LEAFY structures as a guide, it was shown that a region in the LEAFY protein of type I/II allows for binding to sequences with this central region (Figure 2). When this region is mutated (as in the hornwort and algal LEAFYs that bind type III sequences) LEAFY binds to type III sequences. With a molecular understanding of how LEAFY across evolution can bind to different sequences, the original question of how different DNA specificities in a single copy TF could be better addressed. Upon further analysis, the Hornwort LEAFY gene was shown to have structural characteristics of LEAFYs that should allow it to bind both type I/II and type III sequences. Biochemical assays were used to confirm this hypothesis experimentally and showed that this LEAFY is indeed promiscuous in its binding specificity. This seemingly simple finding has profound consequences for our understanding of how TFs could evolve different DNA specificities. Specifically, such an intermediate state could have acted as an ancient evolutionary crossroad through which LEAFY developed into specialised “modern” forms. The reasons for the diversity of modern forms of LEAFY remain elusive, and are the subject of ongoing research.
Principal publication and authors
C. Sayou (a), M. Monniaux (a), M.H. Nanao (b,c), E. Moyroud (a), S.F. Brockington (d), E. Thévenon (a), H. Chahtane (a), N. Warthmann (e), M. Melkonian (f), Y. Zhang (g), G.K.-S. Wong (g,h), D. Weigel (e), F. Parcy (a,i), R. Dumas (a), A Promiscuous Intermediate Underlies the Evolution of LEAFY DNA Binding Specificity, Science 343, 645-8 (2014).
(a) Laboratoire de Physiologie Cellulaire et Végétale (LPCV) [CNRS, UMR 5168/Université Grenoble Alpes/Commissariat à l'Energie Atomique et aux Energies Alternatives, Direction des Sciences du Vivant, Institut de Recherches en Technologies et Sciences pour le Vivant/Institut National de la Recherche Agronomique] Grenoble (France)
(b) European Molecular Biology Laboratory (EMBL), Grenoble (France)
(c) Unit of Virus Host-Cell Interactions, Université Grenoble Alpes–EMBL–CNRS, UMI 3265, Grenoble (France)
(d) Department of Plant Sciences, University of Cambridge (UK)
(e) Department of Molecular Biology, Max Planck Institute for Developmental Biology, Tübingen (Germany)
(f) Botanisches Institut, Lehrstuhl I, Universität zu Köln, Biozentrum Köln (Germany)
(g) Beijing Genomics Institute (BGI)–Shenzhen (China)
(h) Department of Biological Sciences, Department of Medicine, University of Alberta (Canada)
(i) Centre for Molecular Medicine and Therapeutics, Child and Family Research Institute, University of British Columbia (Canada)
References
[1] D. Flot, T. Mairs, T. Giraud, M. Guijarro, M. Lesourd, V. Rey, D. van Brussel, C. Morawe, C. Borel, O. Hignette, J. Chavanne, D. Nurizzo, S. McSweeney, E. Mitchell, J Synchrotron Radiat. 17, 107-18 (2010).
[2] C. Hamès, D. Ptchelkine, C. Grimm, E. Thevenon, E. Moyroud, F. Gérard, J.L. Martiel, R. Benlloch, F. Parcy, C.W. Müller, EMBO J. 27, 2628-37 (2008).
Top image: Structure of the moss LEAFY dimer bound to a type II DNA sequence.