- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Structure of materials
- In situ reduction study of cobalt model Fischer-Tropsch synthesis catalyst
In situ reduction study of cobalt model Fischer-Tropsch synthesis catalyst
Fischer Tropsch (FT) catalysis is an important industrial process for the manufacture of a variety of hydrocarbons and oxygenated hydrocarbons from a mixture of carbon monoxides and hydrogen (in the presence of a catalyst). It generally involves several chemical reactions which can be summarised by:
(2n + 1) H2 + n CO → CnH(2n+2) + n H2O
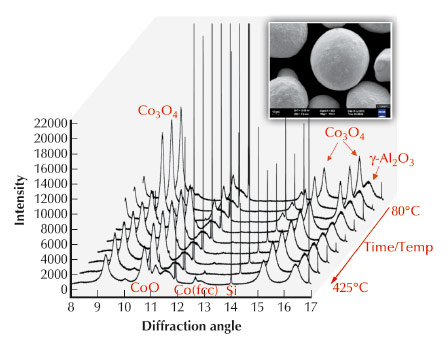
where n is an integer. For cobalt catalysts these reactions occur in the temperature range of 200 - 250°C, and increase in efficiency with pressures [1]. The FT reaction is widely used in industrial plants worldwide in regions of rich natural gas resources to produce liquid fuel products. Cobalt-based catalysts appear to provide the best compromise between performance, life-time and cost for the synthesis of hydrocarbons from CO/H2 mixtures [2]. Cobalt catalysts, like other oxide-based catalysts, undergo a reduction or activation treatment before FT can occur. Cobalt is not used as pure solid, but often supported, i.e. deposited on inert material such as alumina. The insert in Figure 128 shows one such alumina particle (of about 80 µm size) on the surface of which the cobalt oxides are deposited (of sub-micrometre size).
 |
|
Fig. 128: The experimental diffractograms during reduction from 80°C to 425°C, indicating, in particular, the peaks from the Si reference (very narrow), and the evolution of the cobalt oxide to metallic cobalt. |
A fundamental understanding of the exact reduction pathway of supported cobalt oxides and the intermediates can assist in developing improved industrial supported cobalt catalysts. However in situ investigations of the activation process are challenging given the temperature and pressure requirements, and of course the ability to penetrate metallic substances. Thus, the penetration capability and brilliance of hard X-rays from a synchrotron source provide a unique tool for the investigation of chemical processes under in situ conditions and in a time-resolved manner.
Here we present a synopsis of the results from measurements made during in situ reduction of a model Co/alumina catalyst using in situ synchrotron X-ray diffraction at beamline ID31 using both conventional XRD and total scattering (PDF), the latter adds the ability to probe local structures.
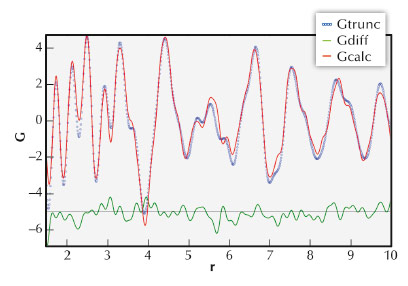
The experiment used a wavelength of 0.3999 Å, which is equivalent to 31.0 keV. The powder diffractograms were collected to a maximum angle of 60°2θ (step size 0.001°2θ) to obtain the resolution required for accurate structure refinement, as well as PDF analysis. Longer counting times were used at higher q-space values (the range of q-space probed was thus 1.00 to 14 Å-1). An inert reference standard, Si, was added to the sample as internal reference to determine the amorphous content in the catalyst during reduction. The reduction was done at ambient pressure in hydrogen with a flow rate of 16 L.g-cat-1.hr-1. The in situ reduction measurements were optimised by measuring 20 minute diffractograms at the following selected temperatures: 80, 160, 200, 240, 260, 280, 320, 360 and 425°C. Six diffractograms were measured at 425°C during a holding time of 2 hours. The heating rate was 10°C/min between measurements, resulting in an average heating rate of 1.6°C/min over the complete reduction. The diffractograms were analysed in Rietveld mode using Topas, and the software package PDFGui. Figure 128 show the diffractograms as collected from 80°C to 425°C. The Si reference peaks are clearly visible, as is the reduction of the cobalt oxide, Co3O4. It is clear that Co3O4 was completely reduced to CoO at around 240°C. The reduction of CoO to cobalt metal was slower with the maximum phase abundance of CoO at 280°C. The gradual decrease of CoO phase abundance coincided with an increase in amorphous content as well as cobalt metal abundance. The quantification of the amorphous content shows the limits of the Rietveld technique, but the total scattering approach provided the additional information contained in the diffuse scattering, Figure 129. Comparison with the ICP-OES results proved that only the total scattering approach can account for all the cobalt in the catalyst.
 |
|
Fig. 129: The PDF fit of the reduced catalyst (Experimental (blue); calculated (red); difference (green)). |
Hard synchrotron X-rays have been successfully used to study the reduction process of Co-based, aluminium supported catalyst in situ. The collected diffractograms allowed the refinement of the different crystalline phases, their structures and volume fractions, as well as probing the local structure (disorder) using PDF refinement. The PDF refinement accounted for all cobalt present in the catalyst sample and showed that after reduction mainly Co metal remained. This is a novel approach to in situ PDF analysis of catalysts containing a mixture of phases.
Principal publication and authors
H.E. du Plessis (a), R.P. Forbes (a), W. Barnard (a), W. Erasmus (a), A. Ferreira (a) and A. Steuwer (b,c), PCCP 15, 11640-11645 (2013).
(a) Sasol technology R&D Pty Ltd, Sasolburg (South Africa)
(b) MAX IV Laboratory, Lund (Sweden)
(c) Nelson Mandela Metropolitan University, Port Elizabeth (South Africa)
References
[1] E. Iglesia, Appl. Cat. A 161, 59–78 (1997).
[2] C.G. Visconti, E. Tronconi, L. Lietti, P. Forzatti, S. Rossini and R. Zennaro, Top Catal 54, 786–800 (2011).



