- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Structural biology
- A transporter caught in the act
A transporter caught in the act
Secondary transporters are membrane proteins that use the energy stored in electrochemical gradients (typically H+ or Na+) to drive antiport or symport of specific solutes. Their function is generally based on an “alternating access” mechanism [1] where the protein switches the orientation between outward- and inward-facing states. This mechanism allows a net transport and accumulation of solute determined by the driving gradient. Over the last 10 years, a wealth of structures of secondary transporters has emerged. However, information as to how transporters actually switch sides and release the driving ion is, in fact, scarce.
Classical examples of secondary transporters include the neurotransmitter-sodium symporters (NSSs) that perform an active re-uptake of molecules such as serotonin, dopamine and γ-aminobutyric acid (GABA). The signalling between neurons is achieved by the release of neurotransmitters into the synaptic cleft from the presynaptic neuron that activates receptors and signal transmission in the postsynaptic neuron. Based on the steep Na+ gradient maintained by the Na+, K+-ATPase, the NSS family transporters perform an active re-uptake to terminate the signal and recycle the neurotransmitters to the presynaptic neuron. It is of particular interest to study the structure-function relationships of this family as they are implicated in various psychological and neurological disorders such as epilepsy, depression and schizophrenia. NSSs are also the key targets for pharmaceuticals including antipsychotics and antidepressants, and for psychostimulants such as cocaine and amphetamine.
Several crystal structures have been obtained of a bacterial member of the NSS family - the amino acid transporter LeuT from Aquifex aeolicus. These have captured the outward-open, sodium-bound state [2], the occluded outward-facing, substrate-bound state [3], and the inward-open apo state [2] and provide snapshots of the beginning and the end of the forward transport cycle. However, the mechanism of intracellular sodium release - where transport becomes coupled to the Na+ gradient - has remained elusive.
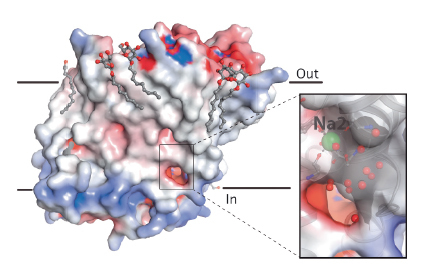
We studied another bacterial model of the NSS family, the multihydrophobic amino acid transporter MhsT from Bacillus halodurans. Two crystal forms of this protein with bound L-tryptophan and sodium ions (diffraction data collected at ESRF beamline ID23-2 and at Diamond Light Source) revealed the long-sought occluded, inward-facing state where sodium and substrate are primed for release to the cytoplasmic environment and highlights both the first steps of solvation of the driving sodium ion and the structural changes that have switched the transporter from an occluded outward- to an occluded inward-facing state. A unique feature was observed: an unwinding at the cytoplasmic end of the fifth transmembrane helix (TM5) allows solvent from the cytoplasm to enter the driving-ion binding site, which also changes from a trigonal pyramidal to an octahedral coordination with a cytoplasmic water molecule as the sixth ligand (Figure 102).
 |
|
Fig. 102: The MhsT inward-facing occluded structure. The electrostatic surface is shown for MhsT. The close-up with a transparent surface shows a solvent-filled, cytoplasmic cavity that reaches the Na2 binding site. Associated detergent molecules are shown as ball-and-sticks, Na+ as green spheres and water molecules as small red spheres (insert). |
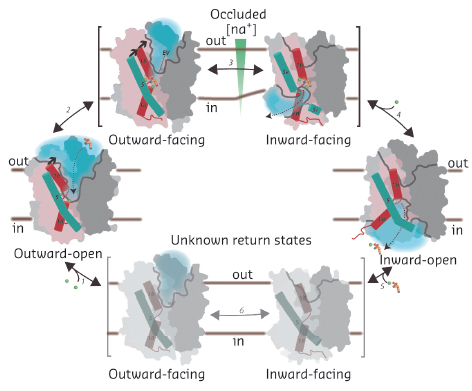
The forward transport reaction can now be described for the NSS family (Figure 103): sodium binding at two sites stabilises the outward-open state [2], and allows substrate binding, which stimulates the transporter to occlude the substrate binding sites [3]. Subsequent closure of an extracellular, hydrophobic vestibule triggers unwinding, at the intracellular side, of TM5. This provides access of cytoplasmic water to the sodium site. This initiates sodium release to the low-sodium intracellular environment, possibly stimulated by a negative membrane potential. After the release of sodium the intracellular cavity opens for complete substrate release to occur [2]. Finally, the transporter switches back to an outward-open state through sodium-free return states, the structures of which are as yet unknown.
 |
|
Fig. 103: Transport mechanism of the NSS family. Na+ binding stabilises the outward-open state (1) and allows the substrate to bind (dashed arrow), triggering substrate site occlusion (2). Closure of a hydrophobic, extracellular vestibule (EV) stabilises the transition to an inward-facing state where TM5 unwinding provides a solvation pathway for the Na2 driving site (3). The helix unwinding provides an opportunity for Na+ to escape to the intracellular low-sodium environment (4). Na+ release from Na2 allows TM5 to reassume a helical structure and TM1a to swing out to release the substrate with Na+ from the Na1 site in the inward-open state. Finally, the transporter switches back to the outward-open state via as yet unknown sodium-free return states (5-6). A scaffold domain of the transporter is shown in dark grey, a bundle domain in pink, half channel openings as grey lines, solvated regions by light blue shadows, TM1 as red and TM5 as cyan cylinders, Na+ ions as green, and amino acid substrate in orange spheres. The membrane is indicated by straight, black lines. |
This new structure in the NSS cycle – caught in the act of exposing sodium to the intracellular environment - provides novel insight into NSS function and, perhaps, into how sodium-dependent transporters generally operate, and brings us closer to a complete picture of their transport mechanism and how this is regulated.
Principal publication and authors
L. Malinauskaite (a), M. Quick (b,c), L. Reinhard (a,d), J.A. Lyons (a), H. Yano (b,e), J.A. Javitch (b,c), P. Nissen (a), Nat Struct Mol Biol. 21, 1006-12 (2014).
(a) Aarhus University (Denmark)
(b) Columbia University College of Physicians and Surgeons, New York (USA)
(c) New York State Psychiatric Institute, New York (USA)
(d) Current address: Karolinska Institutet Stockholm (Sweden) and Karolinska Institutet, Hamburg (Germany)
(e) Current address: National Institutes of Health, Baltimore (USA)
References
[1] O. Jardetzky, Nature 211, 969-70 (1966).
[2] H. Krishnamurthy and E. Gouaux, Nature 481, 469-74 (2012).
[3] A. Yamashita et al., Nature 437, 215-23 (2005).



