- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Structure of materials
- Pushing crystallography across the nucleation barrier
Pushing crystallography across the nucleation barrier
The century-old technique of X-ray diffraction continues to become ever more important for materials characterisation, laying the foundation for many scientific and technological advances in academia and industry alike. The rapid progress in X-ray diffraction methods has made it possible for researchers to routinely identify known materials and solve the structures of previously unknown compounds. The key requirement being that the material exhibits a crystalline long range ordering of the atomic lattice, the prerequisite for Bragg diffraction. Digging into the world of nanoscience, the coherent atomic domains of materials become ever smaller, challenging the condition for Bragg diffraction. This has meant that crystallography was often disregarded in the structural characterisation of amorphous, liquid and extremely nanocrystalline systems. However, this perception is quickly changing, partly owing to the further development and growing use of total scattering experiments and pair distribution function (PDF) analysis [1].
In material science, it is a grand challenge to understand the atomic ordering of materials during synthesis. By knowing the mechanism of crystal formation it may be possible to control it, thereby tailoring desired materials. This is especially a keen interest in the field of solvothermal synthesis [2], where crystalline materials often form from dissolved chemical species or otherwise disordered gel systems. While the evolution of the growing crystalline particles themselves may be followed by diffraction, a barrier is met when trying to understand the local structures existing at the point of nucleation or the stage(s) preceding it.
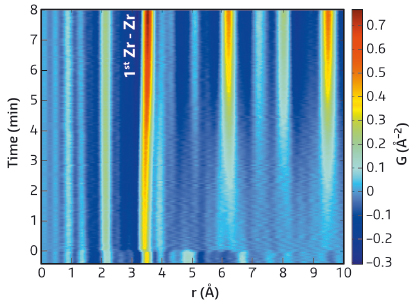
In this study, carried out at beamline ID11, total X-ray scattering in combination with X-ray absorption spectroscopy was used in an attempt to further the understanding of the formation of yttria-stabilised zirconia nanoparticles during solvothermal processing. Under these conditions (methanol-based solution at T = 275°C and P = 250 bar), dissolved Zr4+ and Y3+ species are transformed into crystalline Zr1-xYxO2-δ nanoparticles in a matter of minutes. Visually, the system may be observed to change from a translucent solution to an apparent gel-like structure upon nucleation, steadily densifying with prolonged hydrothermal processing. Therefore, three different structural stages likely exist during different points in this process: 1) pre-nucleation molecular species, 2) a disordered amorphous network and 3) an ordered crystal lattice. The experimental PDF’s representing these different structural stages are shown in Figure 142. Only the third structural stage exhibits Bragg diffraction, the first two stages giving rise to diffuse scattering alone, excluding the use of standard Bragg crystallography. Nonetheless, total scattering clearly proves the existence of distinct local atomic structuring in all three stages spanning further than the first immediate coordination shell.
 |
|
Fig. 142: Local atomic ordering as revealed by total scattering. Time-resolved view of the local structural region (0–10 A°) of the PDF. |
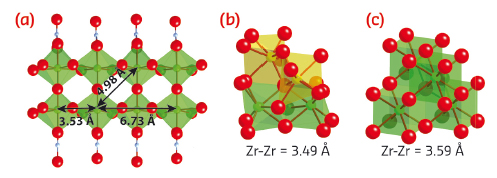
The structures found to best describe these structural stages are shown in Figure 143. The local structure found in the pre-nucleation stage is proposed to consist of polymeric zirconia species, formed during dissolution or present in the solid precursor from the beginning. At the point of nucleation, the data indicate that the polymeric species are rapidly fragmented into smaller species which cluster together and precipitate out as an amorphous network. The atomic network of the precipitate is not entirely randomised despite the apparent chaotic nucleation process, but has a local ordering which may be described as monoclinic in nature. The local bonding environment of the precipitate is therefore not too far from the cubic lattice structure of the end product, yet lacks the correct long range topology. Over time, bond reforming ensures that the local domains order within themselves and in relation to each other thereby forming the desired cubic lattice structure.
 |
|
Fig. 143: Structural stages observed during yttria-stabilised zirconia formation. (a) Proposed solution species structure. (b) Amorphous network structure. (c) Mature cubic crystalline structure of YSZ. |
Numerous simplified models exist to describe crystal nucleation and growth phenomena. The present study provides the first experimental atomistic insight into the very complex chemical processes that take place during crystallisation, and the total scattering technique provides a new approach to truly understanding nanomaterial formation.
Principal publication and authors
C. Tyrsted (a), N. Lock (a), K.M.Ø. Jensen (a,b), M. Christensen (a), E.D. Bøjesen (a), H. Emerich (c), G. Vaughan (d), S.J.L. Billinge (b) and B.B. Iversen (a), IUCr-J 1, 165-171 (2014).
(a) Center for Materials Crystallography, Department of Chemistry, and iNANO, Aarhus University (Denmark)
(b) Department of Applied Physics and Applied Mathematics, Columbia University, New York (USA)
(c) SNBL, ESRF
(d) ESRF
References
[1] T. Egami and S.J.L. Billinge. Underneath the Bragg peaks: structural analysis of complex materials. Vol. 16. Newnes (2012).
[2] K.M. Jensen, C. Tyrsted, M. Bremholm and B.B. Iversen, ChemSusChem 7, 1594-1611 (2014).



