- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2000
- Chemistry
- EXAFS Analyses in Radiopharmaceutical Research: Stability Studies of Technetium(I) Carbonyl Complexes in Solution
EXAFS Analyses in Radiopharmaceutical Research: Stability Studies of Technetium(I) Carbonyl Complexes in Solution
Nowadays, the majority of radiopharmaceuticals used in routine nuclear medicine imaging procedures are 99mTc labelled compounds, and intensive research is in progress to design new 99mTc radiopharmaceuticals. Investigation of Tc and Re carbonyl chemistry has led to an organometallic M(I) aqua ion, [M(CO)3(H2O)3]+ (M = Tc, Re), as a precursor for the preparation of complexes containing the small [M(CO)3]+ moiety. Tc(I) and Re(I) tricarbonyl complexes represent a new and promising approach to low-valent, non-polar and inert compounds. Complexes with N-donor or S-donor chelating ligands coordinated to the M(I) tricarbonyl moiety with high stability in aqueous solution may serve in the design of radiopharmaceuticals [1, 2].
The aim of this work was to investigate the stability of a model Tc(I) dithioether-tricarbonyl complex, Tc1, in aqueous solution and to estimate structural parameters for the dissolved compounds. Owing to the extremely small concentration of the radionuclide present in the radiopharmaceutical preparation (106 108 M), XAS studies as well as most other chemical investigations employ the long-lived isotope 99Tc (K-edge at 21.044 keV). We found that the complex Tc1 is reversibly transformed into the complex Tc1a in aqueous solution (Figure 24).
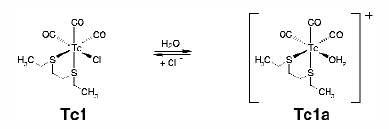 |
Fig. 24: Reversible transformation of the complex Tc1 in aqueous solution.
|
Chemical investigations were performed both at the 99Tc and 99mTc levels. The change of charge of the complexes was determined by electrophoresis. Mass-spectrometric analyses and EXAFS measurements confirmed the composition of the compounds. EXAFS analyses were carried out at BM20 (Rossendorf CRG beamline, ROBL) [3]. 99Tc solutions of Tc1 and Tc1a were prepared by dissolving solid Tc1 in water/methanol and adding chloride or Ag+, respectively. With an excess of sodium chloride the equilibrium (Figure 24) is on the side of the Tc1 complex. If chloride is removed from the equilibrium by precipitation as AgCl, Tc1a is the species in solution. To obtain a satisfactory evaluation of the EXAFS spectra and the Fourier transforms, single-scattering paths including the C, O and S/Cl coordination shells and multiple-scattering paths to model the oxygen atom of the carbonyl group were taken into account (Figure 25). The estimated atomic distances are 1.92 Å (TcC), 2.49 Å (TcS or TcCl), 2.19 Å (TcOaq) and 3.06 Å (TcOCO). The coordination numbers agree with the molecular structure represented in Figure 24. EXAFS analysis of Tc1a shows the same structural parameters for the C, S/Cl and CO coordination shells as Tc1 except that the coordination number for the S/Cl coordination shell is lowered by approximately one. In addition, the detection of 1.2 ± 0.3 oxygen atoms at 2.19 Å proves the substitution of chlorine by water. This can be seen in Figure 25 as a reduction of the magnitude of the second peak in the Fourier transform since electron back-scattering from the oxygen atom is weaker than from the chlorine atom.
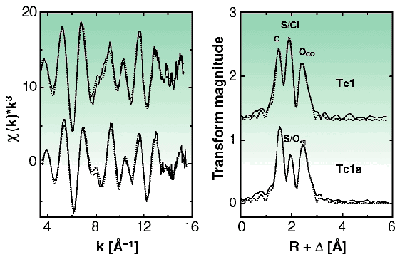 |
Fig. 25: Tc K-edge k 3-weighted EXAFS spectra and the corresponding Fourier transforms of Tc1 and Tc1a (solid line: experimental data, dotted line: fit), transmission mode, sample concentration
|
EXAFS analyses were successfully used to determine structural parameters of Tc(I) tricarbonyl complexes in solution. In connection with chromatographic, electrophoretic and mass-spectrometric methods, it was found that Tc(I) tricarbonyl complexes containing a bidentate thioether ligand are able to react with water by exchange of chlorine.
References
[1] R. Alberto et al., Coordination Chem. Rev., 190-192, 901 (1999).
[2] H.-J. Pietzsch et al., Bioconj. Chem., 11, 414 (2000).
[3] W. Matz et al., J. Synchrotron Rad., 6, 1076 (1999).
Principal Publication and Authors
S. Seifert (a), J.-U. Künstler (a), A. Gupta (a), H. Funke (a), T. Reich (a), C. Hennig (a), A. Roßberg (a), H.-J. Pietzsch (a), R. Alberto (b) and B. Johannsen (a), Radiochim. Acta, 88, 239 (2000).
(a) Research Center Rossendorf (Germany)
(b) University of Zurich (Switzerland)



