- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2002
- Macromolecular Crystallography
- Tandem DNA Recognition by the Two-component Response Regulator PhoB
Tandem DNA Recognition by the Two-component Response Regulator PhoB
Two-component systems are signal transduction pathways widely used by bacteria to sense and respond to environmental changes. In Escherichia coli, PhoR and PhoB constitute a two-component system which controls phosphate uptake. In this system, PhoR is the sensor histidine kinase and PhoB is the response regulator. The former is a transmembrane receptor that senses phosphate levels. When phosphate is low, PhoR autophosphorylates on a histidine residue and transfers the phosphate group to an invariant aspartate located in a conserved regulator domain of PhoB. Upon phosphorylation, the effector domain of PhoB properly binds to the pho box, its specific DNA target sequence, and is able to activate the expression of at least 39 genes implicated mainly in the transport and metabolism of phosphorus compounds [1].
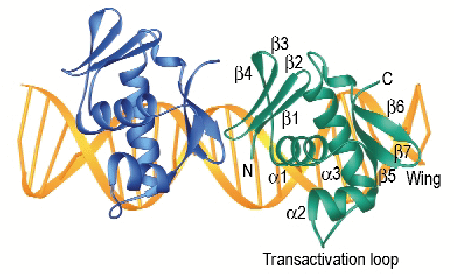
We have used synchrotron radiation to solve the three-dimensional crystal structure of the PhoB effector domain (PhoBE) in complex with pho box DNA at 2.5 Å resolution (Figure 6). The protein presents a winged-helix fold consisting of a central three  -helix bundle packed against two ß-sheets located at each terminus. Helices
-helix bundle packed against two ß-sheets located at each terminus. Helices  2 and
2 and  3 form a modified helix-turn-helix motif where the turn has been replaced by a loop, called the transactivation loop because mutational studies indicate that it is implicated in transcription activation [1]. Finally, the turn located at the C-terminal ß-hairpin constitutes the wing. In the complex crystal structure, two PhoBE protomers are bound in tandem to the same face of the pho box DNA, covering 22 base-pairs and smoothly bending it by 40º. Interestingly, the C-terminal ß-sheet of the upstream protomer (blue in Figure 6) interacts with the N-terminal ß-sheet of the downstream one, directing the head-to-tail DNA binding. DNA recognition elements comprise helices
3 form a modified helix-turn-helix motif where the turn has been replaced by a loop, called the transactivation loop because mutational studies indicate that it is implicated in transcription activation [1]. Finally, the turn located at the C-terminal ß-hairpin constitutes the wing. In the complex crystal structure, two PhoBE protomers are bound in tandem to the same face of the pho box DNA, covering 22 base-pairs and smoothly bending it by 40º. Interestingly, the C-terminal ß-sheet of the upstream protomer (blue in Figure 6) interacts with the N-terminal ß-sheet of the downstream one, directing the head-to-tail DNA binding. DNA recognition elements comprise helices  3, which penetrate the major grooves, and ß-hairpin wings, which interact with the minor grooves. Both ß-hairpin wings compress the minor grooves by inserting Arg219, which is tightly sandwiched between the DNA sugar backbones.
3, which penetrate the major grooves, and ß-hairpin wings, which interact with the minor grooves. Both ß-hairpin wings compress the minor grooves by inserting Arg219, which is tightly sandwiched between the DNA sugar backbones.
 |
|
Fig. 6: Ribbon representation of the PhoBE in complex with the pho box DNA. Two PhoBE protomers are bound in tandem along 22 base pairs of DNA. |
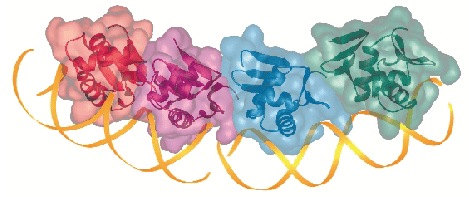
Our findings clarify crucial aspects of the mode of action of PhoB and its homologues, which constitute the OmpR-PhoB family response regulators. Firstly, the protein-DNA complexes are arranged head to tail in the crystal, giving rise to a pseudocontinuous DNA helix, and protein-protein interactions between all neighbouring molecules along this helix are identical. Thus, we have a reliable model of the structure of multiple PhoBE molecules bound to promoters that contain several adjacent pho boxes (Figure 7). Secondly, the transactivation loops protrude laterally, and are thus able to contact the  70 subunit to trigger transcription initiation. Finally, none of the secondary structure elements implicated in DNA binding in PhoB are blocked by the regulator domain in the unphosphorylated and inactive full-length structure of the PhoB counterpart DrrD from Thermotoga maritima [2]. Using this full-length structure as a guide, it is possible to model the interaction between the two domains of full-length, inactive PhoB when complexed to DNA. In this model, severe clashes occur between receiver domains from adjacent PhoB monomers bound in tandem to the DNA. We thus propose that the mechanism for PhoB inhibition is based on steric impediments that prevent the tandem DNA binding needed for transcriptional activation, rather than the blocking of the DNA binding surface by the regulator domain as it occurs in the response regulator NarL, which belongs to a different family [3].
70 subunit to trigger transcription initiation. Finally, none of the secondary structure elements implicated in DNA binding in PhoB are blocked by the regulator domain in the unphosphorylated and inactive full-length structure of the PhoB counterpart DrrD from Thermotoga maritima [2]. Using this full-length structure as a guide, it is possible to model the interaction between the two domains of full-length, inactive PhoB when complexed to DNA. In this model, severe clashes occur between receiver domains from adjacent PhoB monomers bound in tandem to the DNA. We thus propose that the mechanism for PhoB inhibition is based on steric impediments that prevent the tandem DNA binding needed for transcriptional activation, rather than the blocking of the DNA binding surface by the regulator domain as it occurs in the response regulator NarL, which belongs to a different family [3].
 |
|
Fig. 7: Structure of a tetramer of PhoBE bound to two consecutive pho boxes, superimposed with the transparent molecular surface of the protomers, as formed by crystallographic contacts in the crystal structure. |
References
[1] K. Makino, M. Amemura, T. Kawamoto, S. Kimura, H. Shinagawa, A. Nakata and M. Suzuki, J. Mol. Biol. 259, 15-26 (1996).
[2] D.R. Buckler and A.M. Stock, Structure 10, 153-164 (2002).
[3] I. Baikalov, I. Schroder, M. Kaczor-Grzeskowiak, K. Grzeskowiak, R.P. Gunsalus and R.E. Dickerson, Biochemistry 35, 11053-11061 (1996).
Principal Publication and Authors
A.G. Blanco, M. Sola, F.X. Gomis-Rüth and M. Coll, Structure 10, 701-713 (2002).
Institut de Biologia Molecular de Barcelona CSIC (Spain)



