- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2002
- Macromolecular Crystallography
- Structural Basis for Oligosaccharide-mediated Adhesion of Pseudomonas aeruginosa in the Lungs of Cystic Fibrosis Patients
Structural Basis for Oligosaccharide-mediated Adhesion of Pseudomonas aeruginosa in the Lungs of Cystic Fibrosis Patients
The galactose- and fucose-binding (PA-IL and PA-IIL) lectins of Pseudomonas aeruginosa contribute to the virulence of this pathogenic bacterium, which is a major cause of morbidity and mortality in cystic fibrosis (CF) patients via chronic lung colonisation. CF gene mutations increase cell surface fucosylation and CF patients also display modifications in their respiratory and salivary mucins with a higher percentage of sialylated and sulphated oligosaccharides [1]. These cystic fibrosis mucins and cell surface glycoconjugates carry fucose as the terminal sugar residue. Since the P. aeruginosa lectins have been characterised to reveal an outstandingly high affinity of PA-IIL for fucose, they can serve as targets for binding by PA-IIL [2].
Precise three-dimensional knowledge of the lectin sugar-binding site should allow the unusually high affinity to be understood and allow a better design of new anti-bacterial-adhesion prophylactics. With this aim in mind, the crystal structure of the PA-IIL:fucose complex has been determined using the single-wavelength anomalous diffraction (SAD) technique on beamline ID14-1 using the anomalous signal from holmium at 0.934 Å wavelength. Non-isomorphous native data collected on beamline ID14-2 were obtained to 1.3 Å resolution and the holmium structure used as a molecular replacement model for phasing in the asymmetric unit.
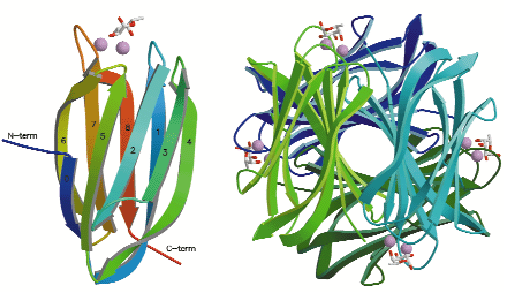 |
|
Fig. 10: Structure of the PA-IIL-fucose complex with stick representation of fucose and calcium ions as space-filling models: (left) Monomer with numbering of ß-strands in agreement with the greek-key motif (1 to 5); (right) The tetramer making up the asymmetric unit. |
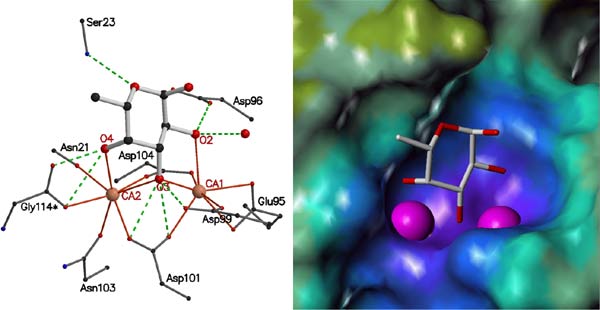
The PA-IIL crystals contain a tetramer with each monomer binding two calcium ions and a fucose molecule (Figure 10). The calcium binding pocket is made up of five acidic groups (Glu95, Asp99, Asp101, Asp 104 and Glu114), the side chain oxygen of Asn103 and a carbonyl oxygen (Asn21) with two of the acid groups (Asp101 and Asp104) bridging between the calcium ions. In each monomer the calcium ions are just 3.75 Å apart and mediate the binding of fucose in a unique and sturdy carbohydrate-recognition mode (Figure 11). The fucose residue itself locks onto both calcium ions, with three of the fucose hydroxyl groups participating in both the coordination of the calcium ions and in hydrogen bonds with acidic groups forming the calcium binding site.
 |
|
Fig. 11: Interactions of PA-IIL with calcium ions and fucose. (a) Stick representation of the amino acids involved in binding. Ca2+ coordination bonds are shown as solid orange lines; hydrogen bonds as dashed green lines. Colour coding: red, oxygen; blue, nitrogen; black, carbon; pink, Ca2+. (b) Electrostatic surface representation of the PA-IIL binding site with Ca2+ as large pink spheres and fucose as a stick model. |
In contrast with most lectins that display only weak affinity for monovalent sugar ligands, the PA-IIL-fucose interaction has an association constant in the micromolar range. The unusual ternary complex formed by the two Ca2+ ions, the side chains of amino acids that coordinate them, and the sugar provides the basis for such strong affinity. In order for sugars to bind to Ca2+ ions as found in the structure of PA-IIL, they must have a particular stereochemical arrangement of three carbohydrate hydroxyl groups. l-fucose, l-galactose, d-mannose and d-arabinose all have the necessary stereochemistry and all of them are recognised by PA-IIL, albeit with diverse affinities [2]. Subsequent modelling studies, based on the fucose complex structure, and binding studies have demonstrated that the preferred ligands of this bacterial lectin belong to the Lewis a series. Such structure-based knowledge could be used for the design of efficient anti-bacterial compounds and, furthermore, the unusually high affinity interaction of this novel binding mode suggests that PA-IIL may be a useful target for oligosaccharide-based therapeutics.
References
[1] Lamblin et al., Glycoconj. J. 18, 661-684 (2001).
[2] N. Garber, U. Guempel, N. Gilboa-Garber and R.J. Doyle, FEMS Microbiol. Lett. 48, 331-334 (1987).
Principal Publication and Authors
E. Mitchell (a), C. Houles (b), D. Sudakevitz (c), M. Wimmerova (b), C. Gautier (b), S. Pérez (b), A.M. Wu (d), N. Gilboa-Garber (c) and A. Imberty (b), Nature Struct. Biol. 9, 918-921 (2002).
(a) ESRF
(b) CERMAV CNRS, Université Joseph Fourier, Grenoble (France)
(c) Bar-Ilan University, Faculty of Life Sciences, Ramat Gan (Israel)
(d) Institute of Molecular and Cellular Biology, Chang-Gung University (Taiwan)



