- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- Structural Biology
- The Structure from Microcrystals of the Cross-beta Spine of Amyloid Fibrils
The Structure from Microcrystals of the Cross-beta Spine of Amyloid Fibrils
Decades of research by medical and biological scientists have established that neurodegenerative amyloid diseases, such as Alzheimer’s, are each associated with a particular protein that converts into fibrillar form. Although these various proteins differ in sequence and three-dimensional structure, their fibrils share common properties: they are ~ 80-150 Å in diameter, highly elongated and unbranched; they bind flat dyes such as Congo Red and display a characteristic birefringence, showing that they harbour an apolar, periodically arrayed environment; they give a characteristic “cross-ß” diffraction pattern, reflecting extended protein (beta) strands, perpendicular to the fibril axis, spaced about 4.8 Å apart. These similarities point to a common structure for the spine of all amyloid fibrils, including the “amyloid-like” fibrils formed from globular proteins by removing them from their native conditions. This common structure resisted detailed elucidation in the past because of the difficulty of extracting atomic-level detail from fibrillar samples, either by NMR or X-ray diffraction.
Our road to the atomic structure of the cross-ß spine had to surmount two major barriers. The first was crystals. We selected the yeast protein Sup35 for X-ray crystallography because extensive studies in other laboratories have shown that its formation of amyloid-like fibrils is the basis of protein-based inheritance and prion-like infectivity. The fibril-forming tendency of Sup35 had been traced to the amino terminus of the protein and from this region we isolated a seven-residue, fibril-forming segment with sequence GNNQQNY [1].
This peptide dissolves in water, and at a concentration of approximately 400 µM, forms amyloid-like fibrils in a few hours. These fibrils display all of the common characteristics of amyloid fibrils. When dissolved at higher concentrations (about 10–100 mM), GNNQQNY forms elongated microcrystals, no larger than ~ 40 µm in length and ~ 1 µm in diameter. Although extremely small, these crystals were well ordered, and were the first truly crystalline form of amyloid-like substances ever discovered.
The second barrier to structure elucidation was how to collect single crystal X-ray diffraction data from such small crystals. Progress came when Dr. Carl-Ivar Branden, the former Scientific Director of ESRF put the UCLA amyloid group in touch with the ESRF team who had developed the microfocus beamline ID13. The diffraction data collected yielded structures for GNNQQNY and for its shorter version NNQQNY [2].
The atomic structure of GNNQQNY revealed some features that were expected, and also some surprises (Figure 78). As expected, each GNNQQNY molecule is an extended beta chain, and each is hydrogen bonded to identical molecules above and below it forming standard parallel ß-sheets. Each sheet is related to a mating sheet by a two-fold axis, and a translation along the fibril axis by one half the inter-strand separation. Surprising to us is that the two sheets are unusually close, with their side-chains intermeshed, forming what we term a ‘steric zipper’. This tight interface between the two sheets is utterly dry, with no water molecules and there are no hydrogen bonds from one sheet to its mate. It is this dry, closely-fitting steric zipper that holds the two sheets together. This same feature is found in microcrystals of other amyloid-forming peptides from proteins associated with human diseases that we have subsequently examined.
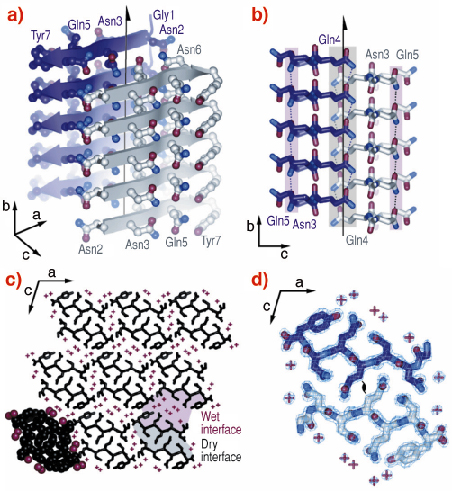 |
|
Fig. 78: Structure of GNNQQNY. Unless otherwise noted, carbon atoms are coloured in purple or grey/white, oxygen in red, and nitrogen in blue. (a) The pair-of-sheets structure showing the backbone of each ß-strand as an arrow, with ball and stick sidechains protruding. (b) The steric zipper viewed down the a axis. The amide stacks of the dry interface are shaded in grey at the centre, and those of the wet interface are shaded in pale red on either side. (c) The GNNQQNY crystal viewed from the top of panel a, along the b axis. Nine pairs of ß-sheets run horizontally. Peptide molecules are shown in black and water molecules are represented by red +. (d) The steric zipper: close-up view of a pair of GNNQQNY ß-sheets from the same view as panel c, showing the remarkable shape complementarity of the Asn and Gln sidechains protruding into the dry interface. 2Fo-Fc electron density is shown, and the position of the central screw axis is indicated. |
The biological implications of the structure of GNNQQNY are: (1) A short peptide of a few (4-7 residues), rather than an entire protein is sufficient to form amyloid-like fibrils. That is, the spine of amyloid fibril has a simpler structure than previously believed; (2) The amyloid state of protein is an unusually dry state; (3) The bond between the two sheets is extended, but formed by weak van der Waals forces. These suggest routes towards developing therapies for terrible diseases such as Alzheimer’s and related amyloidoses.
References
[1] M. Balbirnie, R. Grothe, D. Eisenberg, Proc. Nat. Acad. Sci. USA, 95, 22375-2380 (2001).
[2] R. Nelson, M.R. Sawaya, M. Balbirnie, A. Ø. Madsen, C. Riekel, D. Eisenberg, Nature, 435, 77-778 (2005).
Authors
D. Eisenberg (a), C. Riekel (c), R. Nelson (a), M.R. Sawaya (a), M. Balbirnie (a), A.Ø. Madsen (b,c).
(a) Howard Hughes Medical Institute, UCLA-DOE Institute for Genomics and Proteomics, Los Angeles (USA)
(b) Centre for Crystallographic Studies, Department of Chemistry, University of Copenhagen (Denmark)
(c) ESR



