- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- Structural Biology
- The Structure of Tryptophan 7-Halogenase (PrnA) Suggests a Mechanism for Regioselective Chlorination
The Structure of Tryptophan 7-Halogenase (PrnA) Suggests a Mechanism for Regioselective Chlorination
Chlorinated natural products such as vancomycin and rebeccamycin are widely used in medicine. Although the chemical synthesis of these compounds is known, the agents in clinical use are actually made by fermentation of bacteria. The chlorination of these compounds has long been of interest to chemists and biochemists. This is because the enzymatic transformation which underlies the synthesis must exhibit exquisite regioselectivity. For many years the chlorination of natural products was assumed to rely on haloperoxidases. These are metal based enzymes which utilise hydrogen peroxide to generate a metal bound hypohalous ion. The metal bound intermediate is released as HOCl which then chlorinates substrate. However, this process lacks the regioselectivity seen in many important biologically active chlorinated natural products. Recently, gene sequencing has identified a new class of enzyme that displays regioselectivity and that has been shown to be responsible for the chlorination of almost all bacterial natural products. These enzymes require FADH2 and molecular oxygen for activity and many laboratories have speculated on mechanisms which would explain the regioslectivity observed.
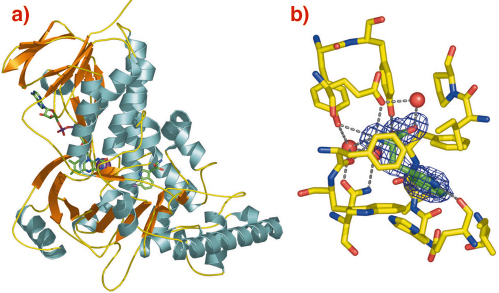
We have determined the native structure of a tryptophan 7-halogenase (PrnA) - a member of the flavin dependent class of halogenases - using Se Met labeling of the protein and diffraction data collected on BM14 (Figure 85a). Structures of PrnA complexed to substrate (tryptophan) and product (7-chloro-tryptophan) were then obtained using data collected on ID14-1 (Figure 85b). The structures show that PrnA can be decomposed into two modules, a flavin binding module and a substrate binding module. The flavin binding module is similar in structure to monooxygenase enzymes and also binds the chlorine atom. The tryptophan binding module is unique. Crucially tryptophan and flavin are separated by 10 Å effectively ruling out any direct transformation of substrate to reaction product.
 |
|
Fig. 85: a) Ribbon diagram of the PrnA-FADH and tryptophan complex. |
Using the data obtained from our structure we were able to design chemical and biochemical experiments to probe the reaction mechanism. Based on these experiments we propose that the enzyme makes HOCl in the flavin module and channels it to the tryptophan module. When at the correct position, HOCl is activated and takes part in electrophilic aromatic substitution. Halogenation using HOCl is well known in nature and organic chemistry, yet it lacks the reactivity and regioselectivity required for biosynthetic pathways. We suggest that with the halogenases described here biology has evolved a remarkable mechanism of generating, activating and controlling HOCl.
Principal Publication and Authors
C.J. Dong (a), S. Flecks (b), S. Unversucht (b), C. Haupt (b), K.-H. van Pée (b) and J.H. Naismith (a), Science, 309, 2216-2219 (2005).
(a) Centre for Biomolecular Sciences, EaStchem, The University, St Andrews (UK)
(b) Institut für Biochemie, TU Dresden (Germany)



