- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2006
- X-ray Imaging and Optics
- Short-term effects of microbeam irradiation on the normal mouse brain microvasculature
Short-term effects of microbeam irradiation on the normal mouse brain microvasculature
Demyelinisation, vascular damage, dementia, white matter radionecrosis, pituitary diseases and hormone depletion occur regularly after high-dose brain irradiation. Such lesions are likely to be the main cause of cognitive dysfunction observed after conventional cerebral tumor radiation treatment with high cumulative doses. The formation of a vasogenic edema following blood brain barrier (BBB) breakdown is thought to contribute to acute radiation damage.
Microbeam radiation therapy (MRT) is an innovative pre-clinical radiation therapy which uses low energetic and non divergent synchrotron wiggler-generated X-rays which are spatially microfractionated to be delivered to the target volume. High flux synchrotron light allows extremely short irradiation times, which avoids broadening of the collimation due to micro-movement of the target. Previous studies have shown that normal cerebral tissues presented particular high resistance to radiation delivered in microbeams [1,2] and that MRT has a curative effect on cerebral tumors implanted in rat brain [2,3]. To explain the sparing effect of microbeam irradiation on normal tissue adjacent to the tumor, it has been hypothesised that non-irradiated endothelial cells between the irradiated zones are able to rapidly repair the damaged microvessels and/or the BBB in the irradiated zones [1,2].
The aim of this study was to determine MRT-induced vascular damage. We focused on the analysis of early effects (12 h – 3 months) of microbeam irradiation on BBB permeability, cerebral blood volume and capillary density for two different MRT-delivered doses: 312 and 1000 Gy.
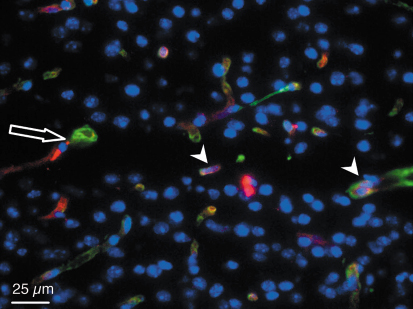
From 12 hours to 3 months after exposure and whatever the dose deposed, there was no difference in the vessel density and in the cerebral blood volume between irradiated and non irradiated cortexes. Endothelial cells remained detectable in the irradiated tissue microslices while neurons and glial cells disappeared during the first week after exposure (Figure 125).
 |
|
Fig. 125: Type IV collagen (green) and platelet endothelial cell adhesion molecule I (red) immunostaining. Horizontal section of a left parietal cortex mouse brain three months after microbeam irradiation with 25 µm-wide microbeams, 211 µm centre-to-centre spacing, skin entrance dose of 1000 Gy. The arrow shows a microbeam path in the motor cortex. An important loss of cell nuclei (blue) can be observed in the microbeam path but immunoreactivity for endothelial cells (arrowhead) remains detectable in the irradiated microslice. |
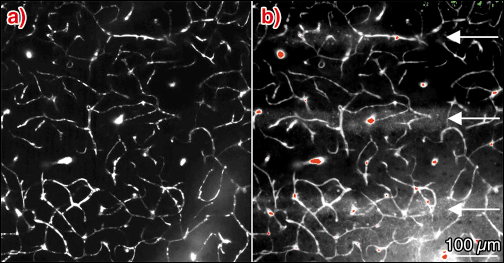
For all time intervals after microbeam exposure and whatever the radiation dose, the FITC-dextran remained in the functional capillary network. From 12 hours to 12 days after irradiation, a diffusion of sulforhodamine B in microbeam stripes was observed for a radiation dose of 1000 Gy. No diffusion was detected 1 month after MRT. For a 312 Gy dose, no leakage of sulforhodamine B in the microbeam stripes of normal brain tissue was detected at any time after MRT (Figure 126).
 |
|
Fig. 126: Z-projection of images acquired in vivo by multiphoton microscopy, 50 to 100 µm below the dura (2 µm steps) in the left parietal cortex of a nude mouse 12 h after 1000 Gy entrance dose exposure. The mouse was injected intravenously with a) 100 µl FITC–dextran (70 kDa) solution and b) 50 µl of a Sulforhodamine B (0.58 kDa) solution, approximately 2 min before microscopy. |
In conclusion, this study shows that microbeam irradiation does not induce severe damage to normal vascular brain tissue. The absence of secondary effects, i.e. weak edema formation, rapid BBB repair and absence of significant changes in cerebral blood volume, indicate that microbeam radiation therapy could be a promising tool in the treatment of malignant brain tumors while preserving healthy tissue.
References
[1] D.N. Slatkin, P. Spanne, F.A. Dilmanian, et al., Proc Natl Acad Sci U S A, 92, 8783-7 (1995).
[2] J.A. Laissue, G. Geiser, P.O. Spanne, et al., Int J Cancer, 78, 654-60 (1998).
[3] F.A. Dilmanian, T.M. Button, G. Le Duc, et al., Neuro-oncol, 4, 26-38 (2002).
Principal Publication and Authors
R. Serduc (a), P. Vérant (b), J.C. Vial (b), C. Rémy (a), E. Brauer (c), A. Bravin (c), J. Laissue (d), B. van der Sanden (a).
(a) INSERM U 594, Grenoble (France)
(b) CNRS UMR 5588, Grenoble (France)
(c) ESRF
(d) Institute of pathology, Bern (Switzerland)



