- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2010
- Structural biology
- Consequences of using X-rays when studying redox cofactors
Consequences of using X-rays when studying redox cofactors
Flavin cofactors are involved in numerous biological processes including light sensing, oxygen activation, electron transfer, and DNA-repair. During these processes the flavin cofactor undergoes subtle conformational changes depending on its state. It is of great importance to understand the connection between flavin conformations and the nature of the chemistry performed.
In the literature, questions regarding flavin structure and function have been addressed many times using spectroscopic methods, theoretical calculations, and X-ray crystallography. The protein data bank (PDB) contains more than 1400 crystal structures containing either a flavin mononucleotide (FMN) or flavin adenine dinucleotide (FAD) cofactor. The redox and protonation states of these cofactors are usually assumed to reflect the situation in the protein crystal prior to X-ray data collection. This is a problematic assumption since redox cofactors are susceptible to react with photoelectrons generated in the crystal by X-rays during data collection. The large distribution of the so-called flavin butterfly bending angle, which is an indicator of flavin red-ox and protonation state, in the PDB-deposited flavoproteins indicate a large diversity of flavin states among these structures [1].
 |
|
Fig. 116: Single-crystal in situ Raman spectra recorded for the flavoprotein NrdI before and after X-ray data collection. |
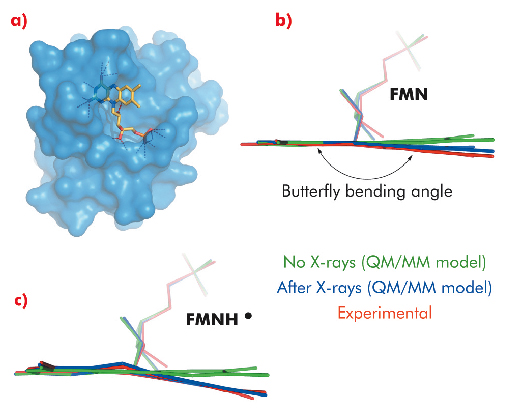
We have solved crystal structures of the flavodoxin-like protein NrdI to 1.12 and 1.15 Å resolution at beamline BM01 using crystals containing the yellow, oxidised FMN (NrdIox) and the blue, neutral semiquinone FMNH• cofactor (NrdIsq), respectively. Before and after X-ray data collection, Raman spectra were collected in situ from both crystals. For both crystals, Raman bands confirmed the initial redox states. However, as shown in Figure 116, dramatic changes in the Raman spectra recorded before and after X-ray data collection were observed. When starting with NrdIox all flavin Raman bands either shift in energy or disappear after X-ray data collection, indicating a change in the flavin state. For NrdIsq all Raman bands decrease in intensity upon X-ray exposure, indicating a one-electron reduction to the fully reduced anionic form FMNH–. These observations coincide with the crystal structures in which the flavin in both models have a butterfly bend deviating from the planarity predicted for FMN and FMNH• (Figure 117).
 |
|
Fig. 117: a) Stick representation of the flavin cofactor bound in a surface pocket of NrdI, b) QM/MM models of flavins in NrdI containing the oxidised FMN cofactor (green), the one-electron reduced (FMN•–) cofactor (blue), and the flavin conformation in the crystal structure of NrdIox (red), c) QM/MM models of flavins in NrdI containing the neutral semiquinone (FMNH•) cofactor (green), the one-electron reduced (FMNH–) cofactor (blue), and the flavin conformation in the crystal structure of NrdIsq (red). In both b) and c) the QM/MM models confirm a one electron reduction of the flavin during X-ray data collection. |
To shed light on our experimental observations, which indicate that the state of the flavin cofactor is influenced by exposure to X-rays, QM/MM models of possible flavin redox and protonation states were built and their geometry optimised. Of the different models containing FMN, FMN•–, FMNH•, FMNH–, or FMNH2, the one-electron reduced forms of the initial states in the crystals, FMN•– and FMNH–, corresponded to the experimentally determined (NrdIox) and (NrdIsq) crystal structures respectively.
The combination of single-crystal in situ Raman crystallography with QM/MM calculations demonstrate the importance of monitoring active site cofactors during X-ray data collection in order to track and characterise eventual radiation damage, which, if unaccounted for, can result in erroneous interpretation of crystal structures [2-5].
Principal publication and authors
Å.K. Røhr, H.-P. Hersleth and K.K. Andersson, Angew. Chem. Int. Ed. 49, 2324-2327 (2010).
Department of Molecular Biosciences, University of Oslo (Norway)
References
[1] T. Senda, M. Senda, S. Kimura and T. Ishida, Antioxid. Redox Signaling 11, 1741-1766 (2009).
[2] H.-P. Hersleth, T. Uchida, Å.K. Røhr, T. Teschner, V. Shünemann, C.H. Görbitz, T. Kitagawa, A.X. Trautwein and K.K. Andersson, J. Biol. Chem. 278, 23372 – 23386 (2007).
[3] H.-P. Hersleth, Y.-W. Hsiao, U. Ryde, C.H. Görbitz and K.K. Andersson, Biochem. J. 412, 257–264 (2008).
[4] H.-P. Hersleth, Y.-W. Hsiao, U. Ryde, C.H. Görbitz and K.K. Andersson, Chem. Biodiversity 5, 2067-2089 (2008).
[5] H.-P. Hersleth and K.K. Andersson, Biochem. Biophys. Acta (2010) (in press doi:10.1016/j.bbapap.2010.07.019).



