- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Soft condensed matter
- From spheres to worms: monitoring transformation kinetics of surfactant micelles using time-resolved SAXS
From spheres to worms: monitoring transformation kinetics of surfactant micelles using time-resolved SAXS
Since their discovery about a century ago, surfactant micelles have been a topic of great interest in both academic and technological research. Surfactant molecules consist of distinct hydrophobic and hydrophilic parts that, in water, lead to aggregation into a wide variety of interesting and highly dynamic nanostructures. The shape and size of the micelles depend on the type of surfactant and conditions such as temperature and solvent composition. Due to the widespread applications ranging from detergents and additives in daily products to advanced applications in biotechnology and nanotechnologies [1], insight into their fundamental behaviour is still highly relevant.
Equilibrium structures of surfactant micelles have been studied extensively, but the pathways of transitions between different morphologies are still not well understood. Micelle kinetics usually takes place in the millisecond and even sub-millisecond regime. This makes it exceedingly difficult to monitor the structural evolution in real time, due to the lack of methods with both nanostructural resolution and millisecond temporal resolution.
Here we use the high brilliance of the ID02 beamline to monitor the kinetics of the transformation from spherical to worm-like micelles induced by adding sodium chloride (NaCl) to an aqueous solution of the archetypical anionic surfactant, sodium dodecyl sulphate (SDS). In aqueous solution SDS forms small spherical or ellipsoidal micelles due to the repulsion between the charged head groups which favours a large surface curvature. By adding salt, the repulsions are screened, and a smaller curvature is favoured, leading to a transition into flexible, “worm-like” cylinders (Figure 31a). Using a stopped-flow apparatus for rapid mixing, combined with the small-angle X-ray scattering (SAXS) setup at ID02, we were able to follow the structural details of the process over time. The time gap between data frames was limited by the detector read-out time, and a stroboscopic-type sampling was employed to capture the rapid kinetic process (Figure 31b). By repeating the mixing sequence several times, varying the delay between the time of mixing and collection of the first data frame, we obtained sampling with only a few milliseconds increment.
 |
|
Fig. 31: a) Transition from spherical to worm-like SDS micelles upon addition of salt. b) Experimental setup where the X-ray pulse is synchronised with the sample mixing. Using a stroboscopic-type sampling, millisecond time resolution was obtained. c) Scattering curves: Intensity vs. modulus of the momentum transfer vector, Q, as a function of time. The data are described using a model for core-shell ellipsoids and worm-like cylinders (lines). |
Figure 31c depicts the scattering curves obtained after mixing a 1% SDS solution with 2 M NaCl (1:1). The intensity rapidly increases, accompanied by a Q-2 behaviour at low Q at later times, demonstrating formation of worm-like micelles. To quantify the results, the data were analysed using a model consisting of a linear combination of ellipsoidal and worm-like core-shell particles [2]. The result, in terms of fraction of surfactant in ellipsoidal micelles and contour length of the worm-like micelles, is plotted in Figure 32.
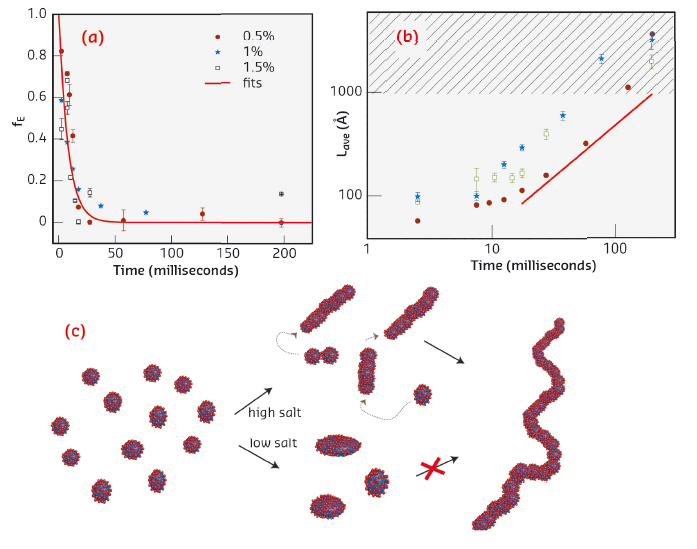 |
|
Fig. 32: Results of the quantitative model fit analysis showing the time dependence of a) the fraction of surfactant in ellipsoidal micelles, and b) the average contour length of the worms. The hatched area indicates the resolution limit of the experiment. c) Schematic illustration of formation of “worm-like” micelles. |
The fraction of ellipsoids approximately follows a single exponential decay, φell(t) ~ exp(–t/τE), with a time constant of about τE = 10 ms (Figure 32a). This decay is followed by growth in the contour length of the worms. Interestingly, the growth approximately follows a linear law Lave ∼ t (Figure 32b). This resembles step-wise growth polymerisation reactions where a linear relationship between mean molecular weight and time is observed [3]. In such reactions, the polymers grow through addition reactions at the chain ends, indicating that a similar mechanism applies here. This suggests a growth mechanism of micelle fusion at the cylinder ends, where the curvature and surface energy is highest (Figure 32c). At a lower NaCl concentration of 1 M, the repulsion between the surfactant head groups is less well screened. The micelles do not undergo a transition into worms, but a slight elongation (within ~10 ms) is observed (Figure 32c).
In conclusion, by time-resolved SAXS, we have been able to shed new light on an old classical surfactant system and obtain insight into morphological transition of micelles. Through a quantitative analysis, we deduced that the kinetic pathway of the transition from spherical to worm-like micelles involves successive fusion events, bearing similarities to classical polymerisation reactions.
Principal publication and authors
G.V. Jensen (a), R. Lund (b), J. Gummel (c), T. Narayanan (c) and J.S. Pedersen (a), Angew. Chem. Int. Ed. 53, 11524–11528 (2014).
(a) Department of Chemistry and Interdisciplinary Nanoscience Center (iNANO), Aarhus University (Denmark)
(b) Department of Chemistry, University of Oslo (Norway)
(c) ESRF
References
[1] D. Attwood and A.T. Florence, Surfactant systems, their chemistry, pharmacy and biology, Chapman and Hall, New York (1983).
[2] J.S. Pedersen and P. Schurtenberger, Macromolecules 29, 7602 (1996).
[3] P.J. Flory, Principles of polymer chemistry, Cornell University Press (1953).



