- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Structure of materials
- Discovery of an unexpected Li-ion battery charging mechanism
Discovery of an unexpected Li-ion battery charging mechanism
The revolution of personal electronics and rise of electronic mobility is made possible by the increasing energy densities of Li-ion batteries. Lithium, which is the third lightest element, is a relatively small ion with a very strong tendency to give away its outer electron. As a consequence, large numbers of lithium ions can be stored at high potential in electrode materials such as LiFePO4, making lithium-ion batteries at least three times more powerful than the traditional nickel-cadmium and nickel metal hydride varieties. One of the main restrictions of today’s Li-ion batteries is the poor kinetics of the phase transitions taking place in the electrodes upon Li-ion insertion and extraction. The impact of defects, particle size and charging rates on the phase transitions in Li-ion battery electrode materials have already been studied intensively, but in situ X-ray diffraction experiments at beamline ID11 have now allowed us to study the chemistry of LiFePO4 batteries during very fast cycling rates for the first time. This effectively made it possible to watch lithium atoms enter and exit the electrodes in real time. We have observed that at high charge and discharge rates, the electrode phase transitions can be bypassed – thereby enhancing the transport of lithium ions through otherwise poorly Li-ion conducting electrode material.
 |
|
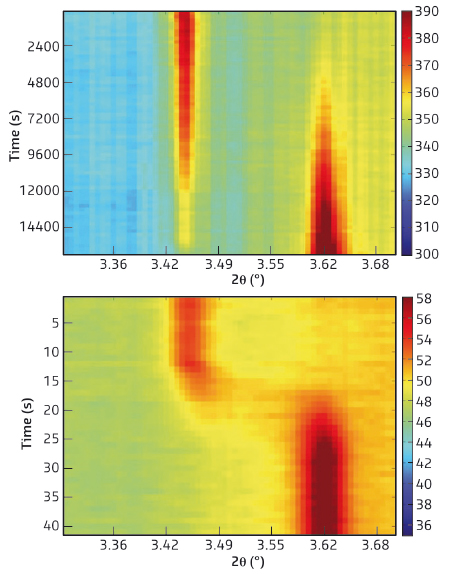
Fig. 131: Top: 2D figure displaying the time evolution of the {200} reflection during slow charging (5 hours). Bottom: 2D figure displaying the time evolution of the {200} reflection during fast charging (1 minute). |
Electrode materials for Li-ion batteries like LiFePO4 store lithium via a reversible first-order phase transition between LiFePO4 and FePO4, which is associated with a reduction in the a-axis lattice parameter. Diffraction during slow charging (5 hours) of the battery demonstrates this phase transition by the gradual disappearance of the {200} reflection of LiFePO4 and the appearance of the {200} reflection of FePO4, shown in the top part of Figure 131. In contrast, during fast charging (1 minute) the {200} reflection gradually shifts from the Li-rich phase position (LiFePO4) towards the Li-poor position (FePO4) showing a considerable intensity between the two end positions, indicating a different phase transition mechanism. The intensity distribution between the FePO4 and LiFePO4 {200} reflections implies that the phase transition progresses via a distribution of lattice parameters, gradually converting LiFePO4 to FePO4. This demonstrates that the dynamic fast charging conditions induce a solid solution transition rather than the thermodynamically predicted first-order phase transition with a phase coexistence of two well defined individual phases.
 |
|
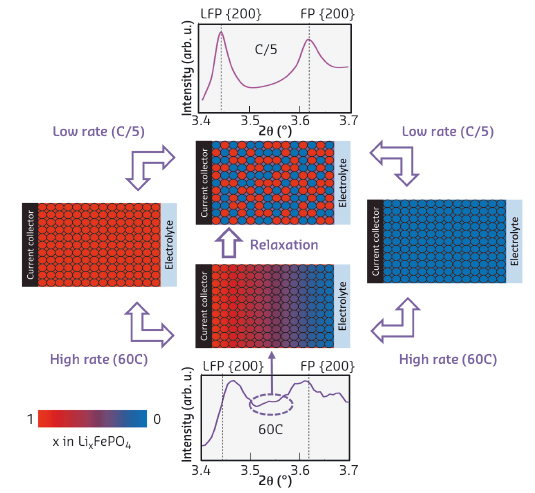
Fig. 132: Model for the phase distribution in LiFePO4 electrodes at high and low rates and upon relaxation. At a low rate of 5 hours (C/5) the electrode is near equilibrium and no significant Li concentrations/overpotentials exist throughout the electrode. At high charge rate (60C corresponding to 1 minute charging) the potential gradient locally causes the bypassing of the first-order phase transition and the appearance of the solid-solution compositions that are observed. |
The in situ fast charging diffraction indicates that only a small fraction of the electrode is actively transforming, consistent with the existence of a Li-ion concentration/potential gradient. The consequence is that the electrode material localised at the gradient experiences an overpotential, absent near equilibrium conditions, that drives the solid-solution transformation. This condition moves like a wave through the electrode as illustrated in Figure 132, transforming the complete electrode. Interrupting the fast charging will allow the system to relax towards equilibrium where, in contrast, the phase transition appears to occur randomly throughout the electrode. From previous research [1], we know that the occurrence of a reacting phase front at high rates indicates that the Li-ion transport in the electrodes limits faster (dis)charging.
Interestingly, the observed bypassing of the first-order phase transition is accompanied by a decrease in internal resistance of the battery. This demonstrates that the solid-state charge transport can be enhanced by the external charge conditions. Against intuition, this may indicate that charging and discharging at higher rates may be beneficial for a battery’s efficiency and cycle lifetime. In addition, we have learned that the key to even faster charging is the Li-ion transport in the electrolyte located in the pores of the electrode, which we can improve by smart design of the electrode morphology. Currently, we are analysing the phase transitions in individual electrode grains in different battery systems under varying charge/discharge conditions, aiming to better understand the nucleation mechanisms, and the development of improved high-performance energy storage in batteries.
Principal publication and authors
X. Zhang (a), M. van Hulzen (a), D.P. Singh (a), A. Brownrigg (b), J.P. Wright (b), N.H. van Dijk (a) and M. Wagemaker (a), Nano Letters 14, 2279-2285 (2014).
(a) Department of Radiation Science and Technology, Delft University of Technology (The Netherlands)
(b) ESRF
References
[1] D.P. Singh et al., Advanced Energy Materials 3, 572-578 (2013).



