- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structure of materials
- Metal-organic framework with novel topology discovered through in situ monitoring of a mechanochemical reaction
Metal-organic framework with novel topology discovered through in situ monitoring of a mechanochemical reaction
A mechanochemical reaction environment has provided the conditions for the formation of an unusual new metal-organic framework possessing new network topology. This discovery was enabled by in situ reaction monitoring via powder X-ray diffraction since the new MOF is a transient intermediate in a multi-step reaction.
The monitoring of mechanochemical milling reactions by X-ray powder diffraction, first developed at the ID15B (now ID15A) high-energy beamline of the ESRF [1], has provided a major breakthrough in the understanding of milling reactions [2], themselves becoming central to solvent- and waste-free chemical transformations. Before the introduction of this technique, in situ monitoring of milling transformations was virtually impossible since such reactions are conducted by rapid oscillation of closed reaction vessels, with the reaction mixture under persistent impact of agitated steel balls and with vessel walls usually made from several millimetres thick steel or tungsten carbide. Application of high-energy X-ray diffraction at ID15B, in combination with reaction vessels manufactured from amorphous and light-atom plastic, now enables in situ monitoring of the evolution of crystalline phases during milling, with time resolution in seconds. The technique has also proven applicable to purely organic materials and the formation of pharmaceutical cocrystals, demonstrating its generality for monitoring a wide range of milling transformations [3].
 |
|
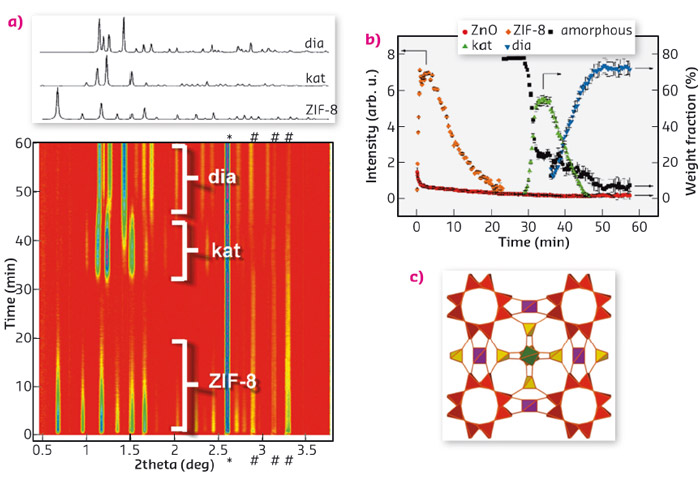
Fig. 34: (a) Time-resolved in situ diffraction experiment exhibiting initial formation of ZIF-8, its amorphisation is evident from the diminishing diffraction signal and subsequent recrystallisation of the amorphous matrix first into the katsenite framework and finally into the diamondoid–topology framework. (b) Time evolution of phases during milling as obtained from Rietveld refinement. (c) Katsenite framework represented by its vertex figure with different colouring for each type of vertex. |
Most recently, we have studied the mechanochemical transformations of the commercially important zeolitic imidazolate framework (ZIF) ZIF-8. In situ monitoring was crucial for the discovery of a new metal-organic framework (MOF), which also revealed a previously unknown tetrahedral framework topology, named katsenite (kat) after A.D. Katsenis, the first author of the article. The discovery of a new ZIF material in a system that was the subject of hundreds of earlier studies revealed, for the first time, that a mechanochemical environment can create conditions for the formation of new phases and structural motifs that cannot be obtained by other conventional methods. Katsenite, itself a polymorph of ZIF-8, was of limited stability under milling conditions and would finally transform to the non-porous diamondoid-topology polymorph of ZIF-8 (Figure 34).
 |
|
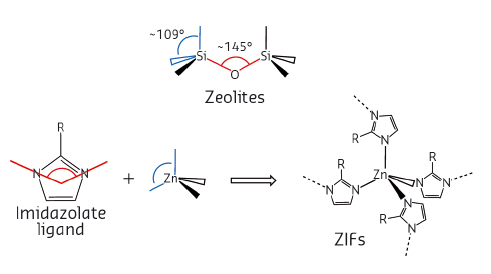
Fig. 35: Similar geometries of valence angles are the reason for coinciding topologies of zeolites and ZIFs. Similar valence angles in zeolites and ZIFs are colour-coded. |
Topologies of most ZIFs can be matched to those of known zeolites. The reasons for this are geometrical, as the tetrahedral metal cation (e.g. Zn2+) mimics the tetrahedral silicon centre of zeolites while the arrangement of nitrogen atoms of coordinating imidazolate linkers mimic the Si-O-Si valence angle characteristic for zeolites (Figure 35) [4,5]. Surprisingly, however, katsenite exhibits a novel topology, never previously seen for zeolites. Its crystal structure was determined from powder X-ray diffraction data to reveal its tetragonal symmetry with four zinc atoms and four imidazolate linkers in the asymmetric unit. The underlying kat-net is unique due to its four non-equivalent 4-c nodes, each adopting a different coordination sequence and point symbol, overall leading to a quadrinodal network based on tetrahedral nodes with transitivity 4463.
A captivating aspect of katsenite is its formation as an intermediate phase in a multi-step process that involves the initial formation of the ZIF-8 sodalite framework by liquid-assisted grinding (LAG) of ZnO and 2-methylimidazole. Its structural collapse into an amorphous material that crystallises into the katsenite structure upon further milling and which, subsequently, arranges into the more stable and completely non-porous diamondoid topology framework during milling. This unusual sequence of events, which is the first example of structural degradation and recrystallisation upon mechanical milling, is highly dependent on using dilute aqueous acetic acid as a catalytic milling liquid additive. While using other additives such as N,N-dimethylformamide (DMF) results in highly crystalline and stable ZIF-8, aqueous acetic acid first catalyses the formation of open ZIF-8 which, surprisingly, collapsed over 20-30 minutes of milling. Importantly, the thus formed amorphous matrix would not always result in the formation of katsenite but would sometimes directly yield the non-porous diamondoid-topology product, serving as the first direct evidence of stochastic, nucleation-related behaviour in mechanochemical synthesis.
Principal publication and authors
In situ X-ray diffraction monitoring of a mechanochemical reaction reveals a unique topology metal-organic framework, A.D. Katsenis (a), A. Puškarić (b), V. Štrukil (a,b), C. Mottillo (a), P.A. Julien (a), K. Užarević (b), M.-H. Pham (c), T.-O. Do (c), S.A.J. Kimber (d), P. Lazić (b), O. Magdysyuk (e), R.E. Dinnebier (e), I. Halasz (b) and T. Friščić (a), Nat. Commun. 6, 6662 (2015); doi: 10.1038/ncomms7662.
(a) Department of Chemistry, McGill University, Montreal (Canada)
(b) Ruđer Bošković Institute, Zagreb (Croatia)
(c) Department of Chemical Engineering, Université Laval (Canada)
(d) ESRF
(e) Max Planck Institute for Solid State Research, Stuttgart (Germany)
References
[1] T. Friščić et al., Nature Chem. 5, 66-73 (2013).
[2] K. Užarević et al., J. Phys. Chem. Lett. 6, 4129–4140 (2015)
[3] I. Halasz et al., Angew. Chem. Int. Ed. 52, 11538-11541 (2013)
[4] X.-C. Huang et al., Chen, Angew. Chem. Int. Ed. 45, 1557 –1559 (2006).
[5] A. Phan et al., Acc. Chem. Res. 43, 58-67 (2010).



