- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2016
- Complex systems and biomedical sciences
- Medical application of synchrotron radiation: towards a new non-invasive method to suppress epileptic seizures
Medical application of synchrotron radiation: towards a new non-invasive method to suppress epileptic seizures
For about a third of patients with epilepsy, current medication is ineffective and, in some cases, surgical resection is the only possible solution. However, this invasive procedure is risky and can only be achieved if the lesion is accessible. Synchrotron microbeam irradiation could offer an interesting alternative for these drug-resistant patients.
At ID17, the biomedical beamline, we have developed a method using microbeams of a few tens of microns that allow the accurate irradiation of specific areas of the brain. Crossing these microbeams at the target region can deposit a sufficient radiation dose for the destruction of certain cells without opening the skull. Previous studies have shown the tolerance of biological tissues to this type of radiosurgery, i.e. the absence of lesions in the immediate vicinity of the microbeam and very few side effects, a significant advantage over current techniques that often present “collateral” damage. The development of the microbeam approach is possible due to the exceptional physical properties of the X-rays produced by the ESRF.
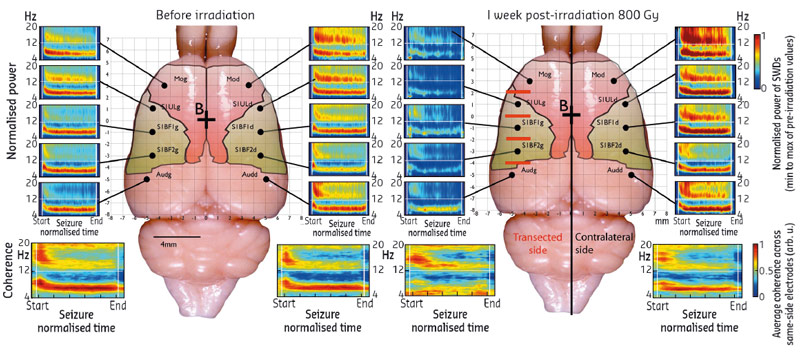
We have demonstrated the possibility of using microbeams to treat some forms of epilepsy during preclinical research. After an initial proof of concept performed on rats [1], we have shown that microbeam irradiation, applied at four different levels of the somatosensory cortex (which generates seizures in this model), has beneficial effects for more than nine weeks (Figure 114).
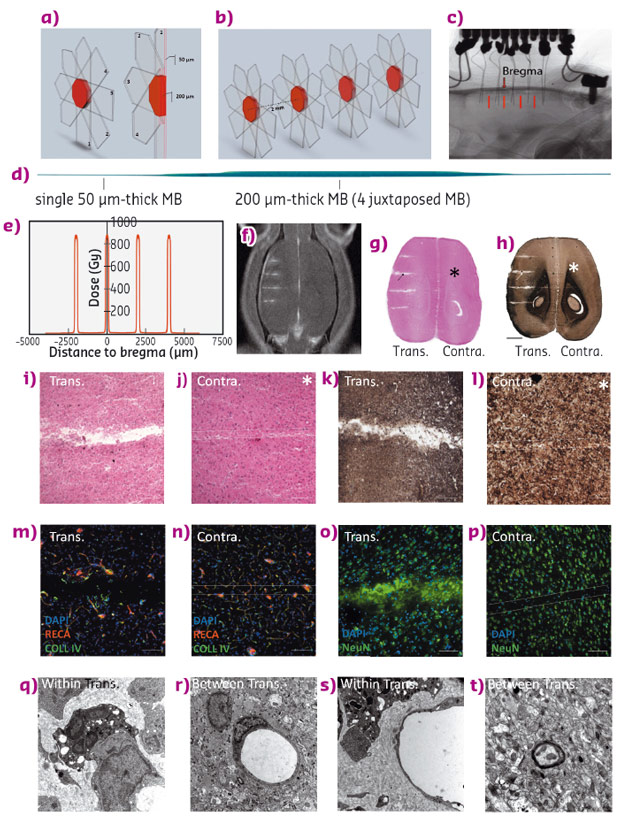
The non-invasive 200 micron wide transections reduce the neuronal connections in the target area, with a resulting reduction in the synchronising capability of neurons, which is monitored by local field potentials. Between radiation zones, magnetic resonance imaging and histological analyses showed that the tissue is not altered and behavioural tests have shown that animals retain normal locomotion and motor coordination. Figure 115 shows the irradiation geometries, a lateral dose profile, MRI images together with histological sections using various staining techniques.
 |
|
Fig. 115: Synchrotron X-ray transections. Irradiation parameters and tissular effects. a), b) 3D Microbeam interlacement; transparent arrows denote the trajectory of each microbeam. Numbers represent the sequence of their delivery. The interlaced volume (transection) is shown in red. c) X-ray image of a GAERS brain with two rows of 5 implanted electrodes. Transections are indicated by red bars. d) The scan of a Gafchromic™ film shows 4 microbeams, interlaced to create a 200 μm wide, octahedric irradiated transection. e) Radiation dose deposition profile for the 4 transections. Estimated peak dose in the transection 820 Gy, entrance dose of a single microbeam 800 Gy. f) Gadolinium contrasted T1 MR image of a rat after 4 transections. g) to t) Horizontal brain sections 2 months post-irradiation, haematoxylin and eosin (g), myelin stain (h); scale: 2 mm. Microphotographs of cortical transections (i, k) show a tissue gap (i), and myelin loss (k), while the path of a single microbeam in the contralateral hemisphere shows only loss of cell nuclei (j and l); scale: 200 µm). Immunolabelling of brain vasculature (RECA and Type IV Collagen (m, n) and neurons (NeuN (o, p)) in transected (m, o) and contralateral (n, p) hemispheres (scale: 50 µm). Ultrastructural changes, as described in the text, within (q, s) and between transections (r, t). (TEM, magnification 12,000). |
This new study confirms the interest of using synchrotron radiation to reach epileptic foci where conventional surgery is too risky. It constitutes a new therapeutic approach that needs to be confirmed in other models before considering clinical studies. The method can also be applied with very fine transections (50 microns) to reach precise brain regions and it can also find applications in the study of brain connectivity in neuroscience.
Principal publication and authors
Synchrotron X-ray microtransections: a non invasive approach for epileptic seizures arising from eloquent cortical areas, B. Pouyatos (a,b,c), C. Nemoz (d), T. Chabrol (e), M. Potez (b), E. Bräuer (d), E. Renaud (f,g), K. Pernet-Gallay (a,b), F. Estève (e), O. David (a,b), P. Kahane (a,b,h), J.A. Laissue (i), A. Depaulis (a,b) and R. Serduc (e), Scientific Reports 6, 27250 (2016); doi: 10.1038/srep27250.
(a) Inserm, U1216, Grenoble (France)
(b) Université Grenoble Alpes, Grenoble Institut des Neurosciences, GIN, Grenoble (France)
(c) Synapcell S.A.S, La Tronche (France)
(d) ESRF
(e) Grenoble Alpes EA RSRM, Grenoble (France)
(f) CNRS;CE2F PRIM UMS3537, Aix-Marseille Université (France)
(g) Aix Marseille Universite, Centre d’exploration Fonctionelle et de Formation (France)
(h) CHU Grenoble Alpes, Grenoble (France)
(i) University of Bern (Switzerland)
References
[1] B. Pouyatos et al., Neurobiology of Disease 51, 152–160 (2013).
[2] F. Studer et al., Phys Med 31, 607–614 (2015).