- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2017
- Complex systems and biomedical sciences
- Revealing the interplay between structure and electronic coupling in donor/acceptor systems for organic photovoltaics
Revealing the interplay between structure and electronic coupling in donor/acceptor systems for organic photovoltaics
A comprehensive study on the charge-transfer effect in small-molecule organic semiconductor thin films has been performed. Using a combination of structural and optical methods, it was established that strong intermolecular coupling in molecular co-crystals results in new electronic properties, in particular a strong anisotropic charge-transfer transition.
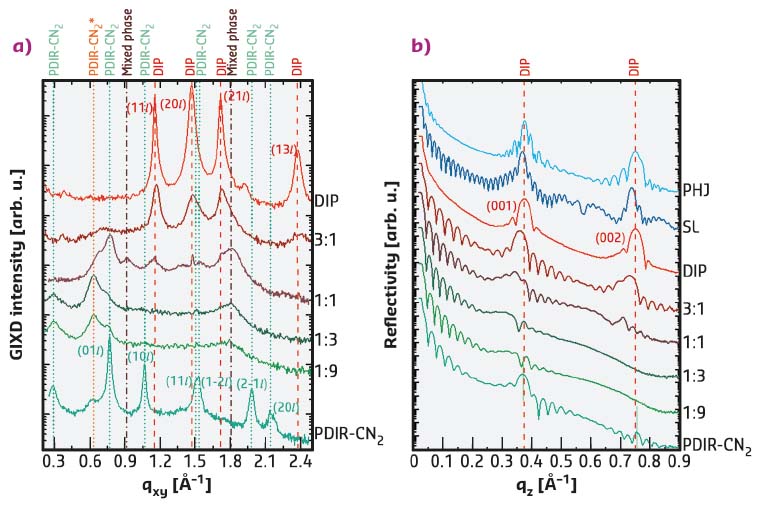
One of the fundamental processes in devices based on organic semiconductors such as organic solar cells and organic light-emitting diodes is charge-transfer (CT) between electron donor (D) and electron acceptor (A) molecules at interfaces or in molecular mixed systems. A and D form a CT complex, where the energy levels of the ground and excited states depend on the overlap of the D/A wave functions, and the electronic transition to the excited state is accompanied by partial or integer charge transfer. An important question for the efficiency is the anisotropy of the CT state, i.e. differences in the electronic coupling in-plane vs. out-of-plane. The present study addresses this by complementary use of structural and spectroscopic tools. The key to success was the careful preparation of the different layer sequences and compositions, with high-precision characterisation at beamline ID10 using surface X-ray scattering (grazing-incidence X-ray diffraction (GIXD) and reflectivity (XRR), see Figure 58).
 |
|
Fig. 58: a) GIXD data and b) XRR data allow the mixed phase formation to be observed. All spectra are vertically offset for clarity. |
Two perylene derivatives, DIP and PDIR-CN2, were found to be an excellent model system to study CT effects in organic semiconductors. Three different architectures were chosen: a planar heterojunction (PHJ), a two-component mixed bulk-heterojunction film (BHJ) with different mixing ratios, and a superlattice (SL) consisting of 20 alternating monolayers as an intermediate case. The two compounds were characterised regarding structural organisation of the molecules during simultaneous or consecutive growth by organic molecular beam deposition (OMBD), as this is crucial for the formation of D/A interfaces. The result demonstrates a high miscibility between DIP and PDIR-CN2 molecules, leading to the formation of a new mixed phase polymorph. Apparent changes in lattice parameters show the reorientation of D and A molecules thus creating face-to-face stacking with molecular planes parallel to each other. Despite weak crystallinity in the in-plane direction, mixed films in the absence of phase separation exhibit preferential D/A stacking, which is expected to enable CT state formation in this direction.
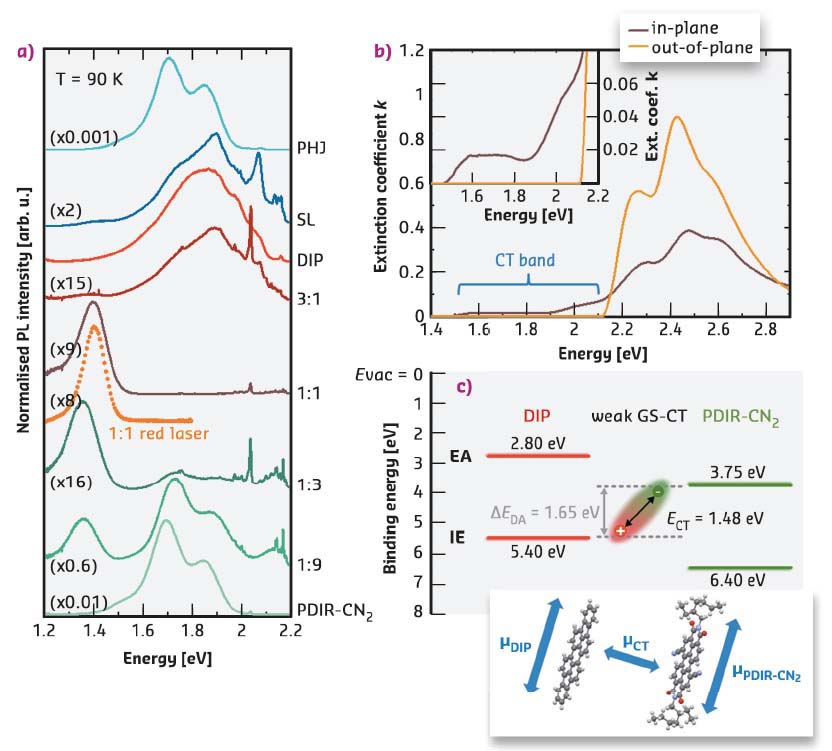
Spectroscopic methods were used for the characterisation of the electronic coupling (Figure 59). In Figure 59a a new emission band, emerging around 1.4 eV, was assigned to radiative recombination processes from the charge-transfer state between DIP and PDIR-CN2 molecules apparently accompanied by enhanced non-radiative recombination, that was interpreted from its strong photoluminescence quenching. The CT state can be excited directly, avoiding transfer from higher-energy molecular excitons, confirmed by using a red laser. Regarding the layered samples, the PHJ exhibits strong emission dominated mainly by PDIR-CN2 excitonic recombination. Emission of the SL contains features of both components, and only weak emission features from the CT states can be observed.
 |
|
Fig. 59: a) Photoluminescence spectra. b) Anisotropic components of the extinction coefficient of the 1:1 film. The inset zooms into the range of 1.4−2.2 eV. c) Schematic energy-level diagram for the mixture DIP/PDIR-CN2 (top) and orientations of transition dipole moments (bottom). |
The absorption spectrum of the equimolar mixed film clearly reveals a broad band at photon energies below the optical gaps of the pristine materials (1.5−2.1 eV, Figure 59b), which originates from direct excitation of CT states. From the overlap of the absorption and emission spectra, the dominant CT state energy can be deduced. The energy-level diagram of the resulting CT complex is depicted in Figure 59c, where the energy gap ΔEDA and the CT state energy give a CT exciton binding energy of about 0.2 eV. Spectroscopic ellipsometry allows the two anisotropic components of a 1:1 film to be distinguished as the CT band is only visible in the in-plane direction, which is the direction of the π−π interaction. Intermolecular D/A interactions are directly related to the overlap of their frontier orbitals, which is maximal in π−π stacking or face-to-face geometry (perpendicular to the molecular backbones). Therefore, the presented CT is stronger in mixed films, where molecule reorientation facilitates orbital overlap, in contrast to planar interfaces (PHJ and SL) with almost exclusively edge-on-edge stacking. The anisotropic character of the electronic transition to the excited CT state polarised in the in-plane direction is sketched in Figure 59c (bottom). Thus, π−π stacking is a prerequisite for efficient intermolecular coupling, while the strong intermolecular interaction itself seems to play a role as one of the driving forces for molecular (re)arrangement.
Principal publication and authors
Evidence for anisotropic electronic coupling of charge transfer states in weakly interacting organic semiconductor mixtures, V. Belova (a), P. Beyer (b), E. Meister (c), T. Linderl (c), M.U. Halbich (d), M. Gerhard (d), S, Schmidt (c), T. Zechel (c), T. Meisel (b), A.V. Generalov (e), A. S. Anselmo (f), R. Scholz (h), O. Konovalov (g), A. Gerlach (a), M. Koch (d), A. Hinderhofer (a), A. Opitz (b), W. Brütting (c) and F. Schreiber (a), J. Am. Chem. Soc. 139, 8474−8486 (2017); doi: 10.1021/jacs.7b01622.
(a) Institut für Angewandte Physik, Universität Tübingen (Germany)
(b) Department of Physics, Humboldt-Universität zu Berlin (Germany)
(c) Institute of Physics, Experimental Physics IV, University of Augsburg (Germany)
(d) Faculty of Physics and Material Sciences Center, Philipps-Universität Marburg (Germany)
(e) Max IV Laboratory, Lund University (Sweden)
(f) Helmholtz-Zentrum Berlin für Materialien und Energie GmbH, Berlin (Germany)
(h) Dresden Integrated Center for Applied Physics and Photonic Materials (IAPP), Technische Universität Dresden (Germany)
(g) ESRF



