- Home
- News
- Spotlight on Science
- X-rays reveal single...
X-rays reveal single catalyst nanoparticle at work
08-10-2021
X-ray diffraction imaging at ID01 has been used to observe a single catalyst nanoparticle at work, revealing for the first time how the chemical composition of the surface of an individual nanoparticle changes under reaction conditions. This study marks an important step towards a better understanding of industrial catalytic materials.
Share
Catalysts are materials that promote chemical reactions without being consumed themselves. Today, catalysts are used in numerous industrial processes, from fertiliser production to manufacturing plastics. Because of this, catalysts are of huge economic importance. A well-known example is the catalytic converter installed in the exhaust systems of cars. These contain precious metals such as platinum (Pt), rhodium (Rh) and palladium (Pd), which allow highly toxic carbon monoxide (CO) to be converted into carbon dioxide (CO2), and reduce the amount of harmful nitrogen oxides (NOx).
Despite their widespread use and great importance, many questions remain regarding how various catalysts work. It is difficult to study real catalysts while in operation as the catalysts are typically used in the form of tiny nanoparticles in order to make the active surface as large as possible, and the changes that affect their activity occur on their surface.
A new technique for labelling individual nanoparticles and thereby identifying them in a sample has been developed by a team of researchers from DESY NanoLab. Nanoparticles of a PtRh alloy were grown on a SrTiO3 substrate and one specific particle, Pt60Rh40, which was around 100 nanometres in diameter, similar to the particles used in a car’s catalytic converter, was selected for characterisation.
Coherent X-ray diffraction imaging (CXDI) was carried out at beamline ID01, enabling the team to create a detailed image of the nanoparticle, and to measure the mechanical strain within its surface (Figure 1). Since the surface strain is related to the surface composition, in particular the ratio of platinum to rhodium atoms, the strain was computed as a function of surface composition. By comparing the observed and computed facet-dependent strain, conclusions can be drawn concerning the chemical composition at the particle surface.
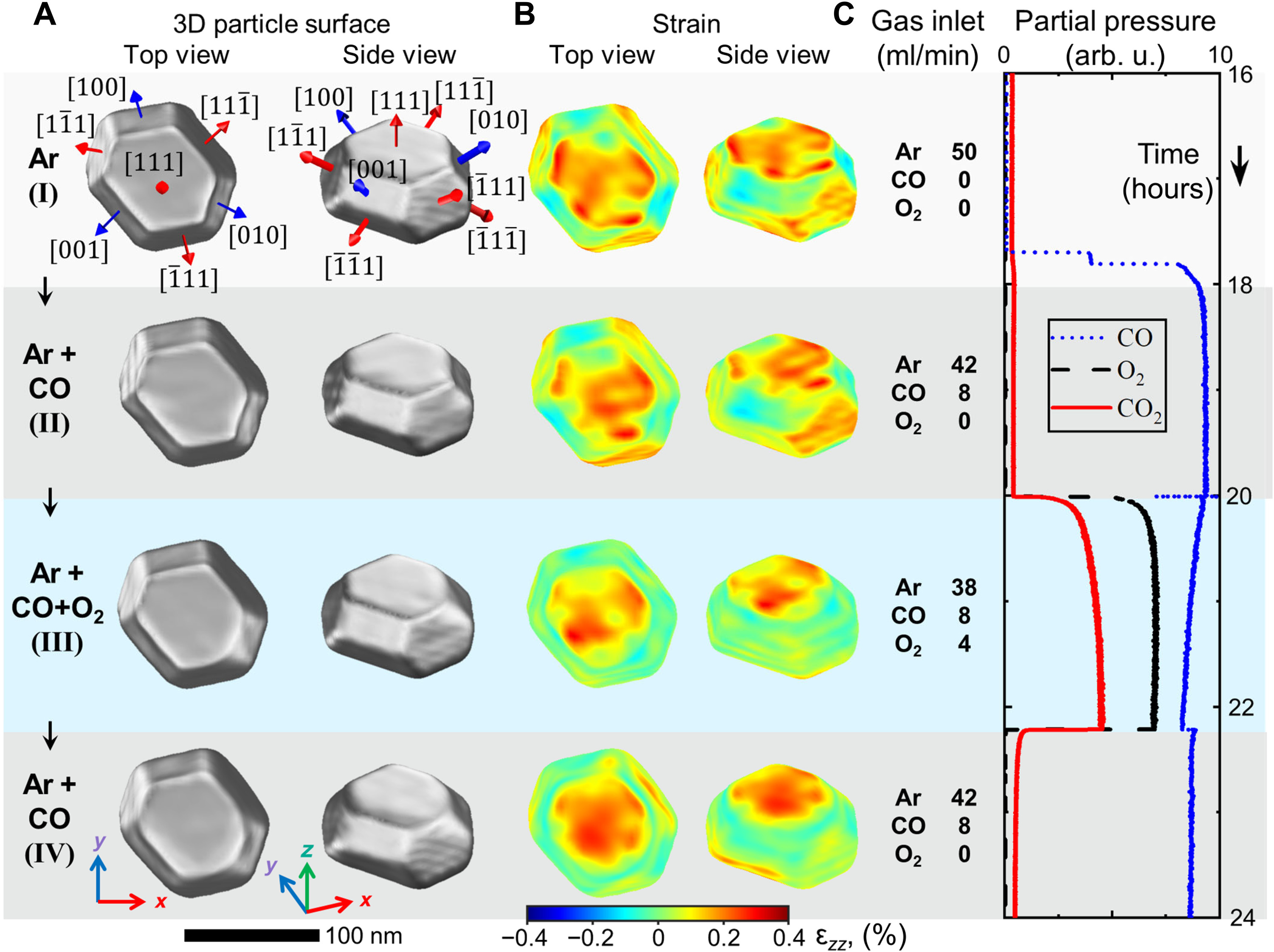
Fig. 1: Nanoparticle shape and surface strain for different gas conditions. a) Top and side views of the reconstructed nanoparticle and (b) strain field ϵzz at the nanoparticle surface for gas conditions (I to IV). The position of the surface is defined as a cut at 55% of the reconstructed crystalline electron density from its maximum value. c) Inlet gas composition and mass spectrometer signal during the experiment: CO (blue), O2 (black), and CO2 (red).
When the nanoparticle is grown, its surface consists mainly of platinum atoms, as this configuration is energetically favoured. However, the shape of the particle and its surface strain were studied under different gas conditions, including the operating conditions of an automotive catalytic converter, created by heating the particle to around 430 degrees Celsius and allowing carbon monoxide and oxygen molecules to pass over it. Under these reaction conditions, the rhodium inside the particle becomes mobile and migrates to the surface because it interacts more strongly with oxygen than the platinum. This is also predicted by theory.
As a result, the surface strain and the shape of the particle change. A facet-dependent rhodium enrichment takes place, whereby additional corners and edges are formed. The chemical composition of the surface, and the shape and size of the particles have a significant effect on their function and efficiency. It is still not completely understood how these are connected and how to control the structure and composition of the nanoparticles. However, CXDI made it possible to detect changes of as little as 0.1 in a thousand in the strain, corresponding to a precision of about 0.0003 nanometres in this work.
This work shows that, for the first time, the details of the structural changes in such catalyst nanoparticles can be observed while in operation. This is a major step forward in the understanding of an entire class of catalysts, namely in all reactions that make use of alloy nanoparticles. This investigation is also an important step towards analysing industrial catalytic materials. Until now, model systems have had to be grown in the laboratory in order to conduct such investigations. Future work aims to study individual particles that are ten times smaller, in real catalysts and under reaction conditions.
Principal publication and authors
Single Alloy Nanoparticle X-Ray Imaging during a Catalytic Reaction, Y.Y. Kim (a), T.F. Keller (a,b), T.J. Goncalves (c), M. Abuin (a), H. Runge (a), L. Gelisio (a), J. Carnis (a), V. Vonk (a), P.N. Plessow (c), I.A. Vartanyants (a,d), A. Stierle (a,b), Sci. Adv. 7, 40 (2021); https://doi.org/10.1126/sciadv.abh0757
(a) Deutsches Elektronen-Synchrotron (DESY), Hamburg (Germany)
(b) University of Hamburg, Physics Department, Hamburg (Germany)
(c) Institute of Catalysis Research and Technology, Karlsruhe Institute of Technology, Leopoldshafen (Germany)
(d) National Research Nuclear University MEPhI, Moscow (Russia)



