- Home
- News
- Spotlight on Science
- X-ray nanotomography...
X-ray nanotomography reveals 3D microstructure of graphite anodes for lithium-ion batteries
17-05-2023
X-ray phase-contrast nanotomography provides unique capabilities for in-depth 3D internal structural analysis of the complex architecture of battery electrodes. In this work, this nanoimaging technique was used at beamline ID16B to highlight the impact of graphite electrode calendering and particle coating on the microstructure of a graphite electrode, and its related electrochemical performance.
The optimisation of battery electrode architecture is a key aspect of improving battery performance, provided that precise characterisation of the complex battery microstructure is possible. In this work, X-ray nanotomography [1] was used at beamline ID16B [2] to obtain high-resolution images of the microstructure of graphite battery electrodes, providing 3D analysis and thorough quantification of the electrode/particle inner structure and porosity at the nanoscale.
A crucial step in the production of battery-grade natural graphite for lithium-ion batteries is the spheroidisation process, in which the graphite flakes are mechanically shaped via sophisticated milling techniques into nearly spherical particles to decrease their surface area and to increase the material density. However, the low yield (30-50%) of this process results in a large quantity of wasted graphite fines that are not suitable for use in lithium-ion batteries due to their small particle size [3]. A method was devised to recycle waste graphite fines via a re-agglomeration process followed by a petroleum pitch coating in order to obtain aggregated graphite particles with sound mechanical strength and battery-suitable size to be used for electrode preparation. A compression step called ‘calendering’ was applied to the electrode to reduce its thickness and consequently increase its volumetric capacity.
Click image to enlarge
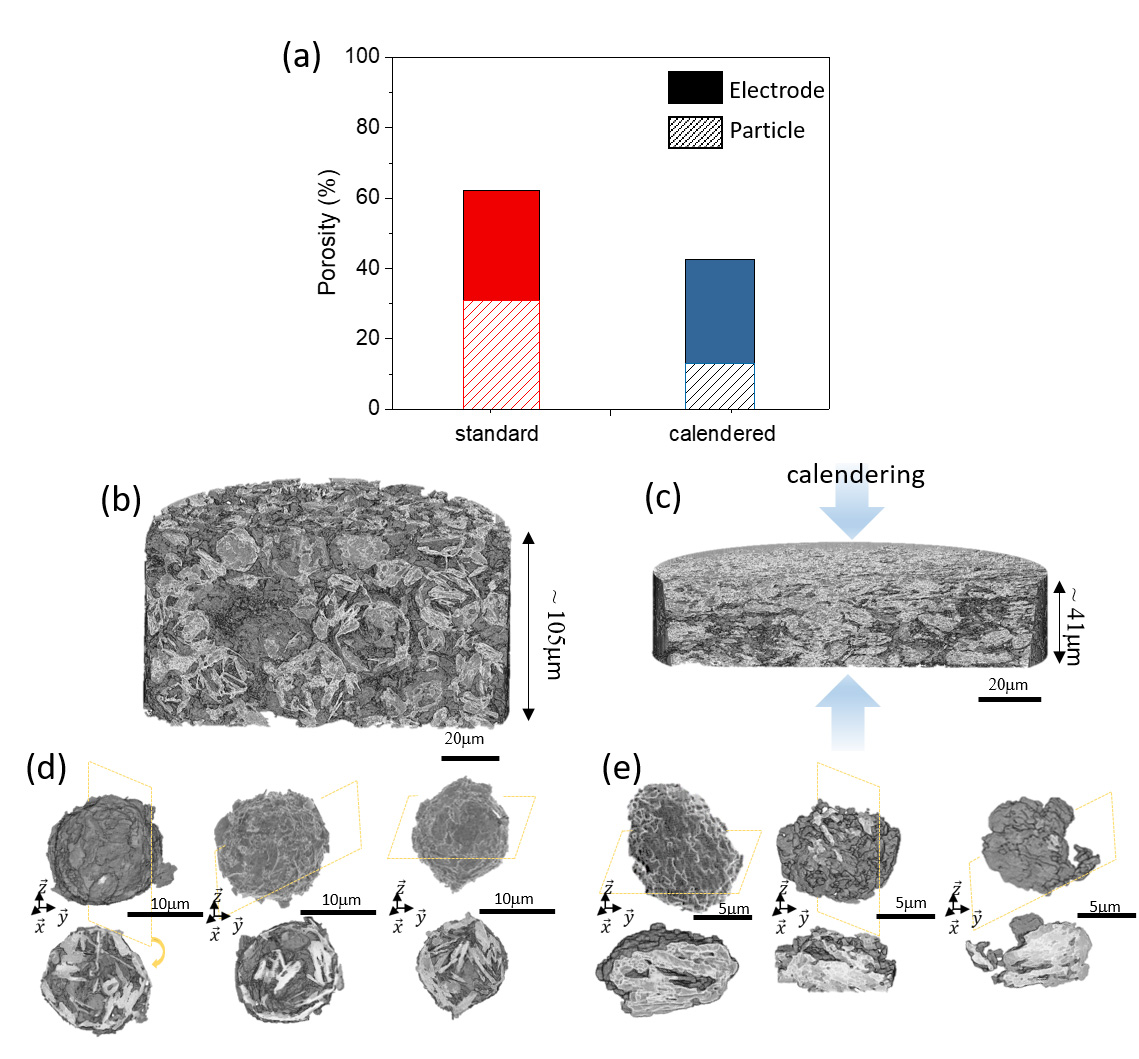
Fig. 1: Electrode and particle porosity evolution with calendering in terms of (a) pore volume fraction and (b-e) microstructure. 3D rendering views of the (b) non-calendered and (c) calendered electrodes and (d,e) corresponding isolated graphite aggregated particles (with cross-section images).
X-ray nanotomography measurements carried out at beamline ID16B provided important microstructural details of the electrode-representative volumes (128 × 128 × 108 µm3 with 50 nm voxel size), along with statistical analysis of ~500 particles imaged in a single measurement (Figure 1a). Data acquired on non-calendered and calendered pristine electrodes (Figures 1b and c) show that higher electrode density could be reached by calendering the electrode, without considerably affecting the active material accessibility through diffusion in the pore network. Despite the considerable morphological changes, no clear agglomerate fractures were observed, and particle integrity was preserved as individual agglomerate particles could still be distinguished (Figures 1d and e). This highlights the fact that structural integrity is maintained from the electrode scale down to the particle level, and that the calendering process does not compromise the electrochemical performance.
Click image to enlarge
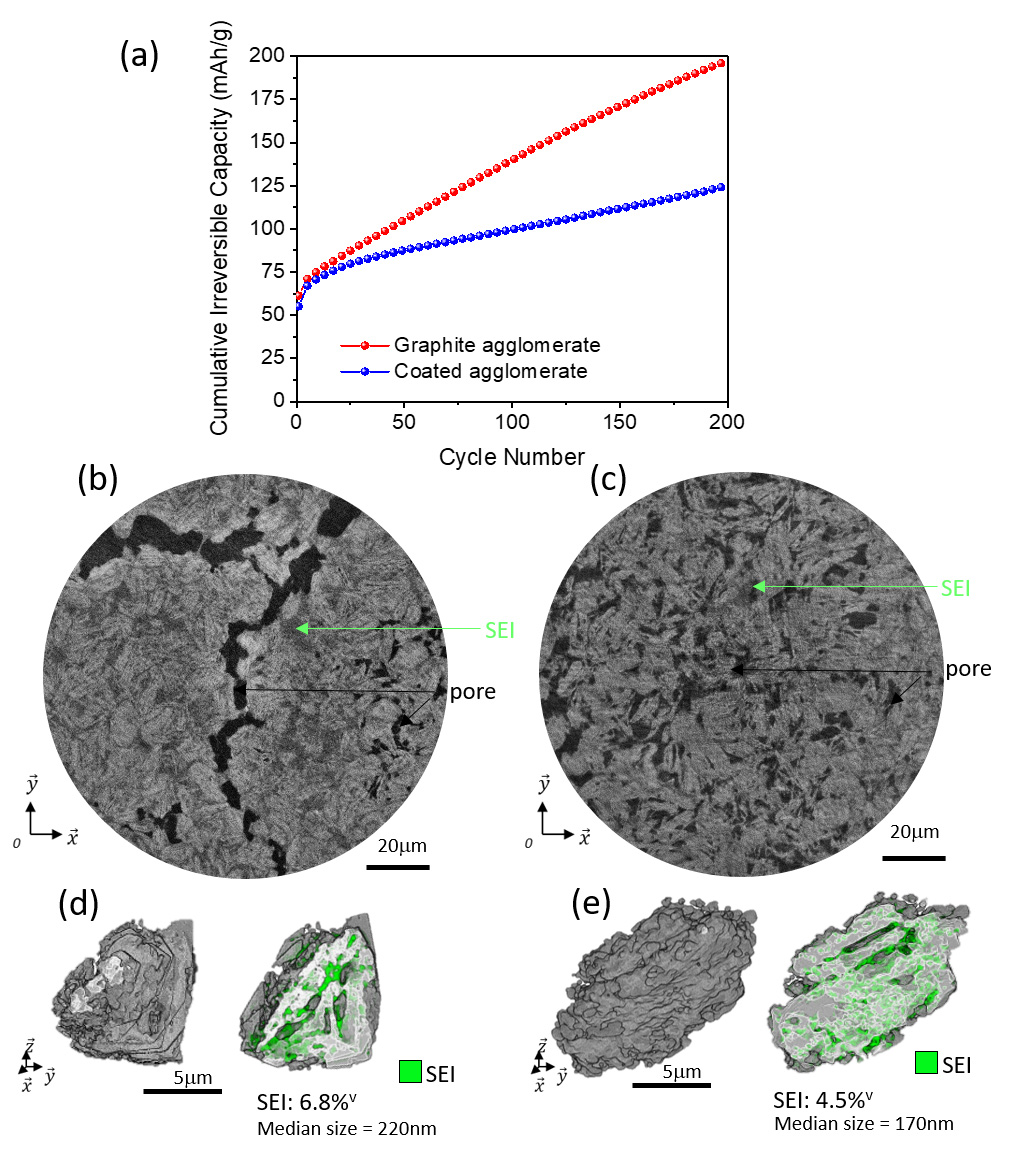
Fig. 2: Evolution during cycling of (a) cumulative irreversible capacity and electrode/particle microstructures for (b,d) uncoated and (c,e) coated graphite agglomerates after 200 cycles.
Complementary post mortem measurements performed on dismantled electrodes (retrieved after 200 cycles) compared graphite anodes of non-coated aggregated graphite particles with pitch-coated ones. Phase-contrast images revealed the presence of an important Solid Electrolyte Interface (SEI) decomposition layer formed during cycling (Figures 2b and c). At the electrode scale, the volume fraction of bulk SEI formed was larger for the non-coated material by nearly a factor of two. At the particle level, the total volume of inner decomposition layer formed in the graphite aggregates was reduced by nearly a third on the pitch-coated particles (Figures 2d and e). The striking difference between the two formulations highlights the benefits of the coating step for reducing the continuous SEI formation both on the surface of the active material and inside the aggregates. The image analysis directly supports the important difference measured in terms of cumulated irreversible capacity, defined as the sum over cycles of the excess lithiation capacity versus delithiation capacity at each cycle, which is mainly related to the SEI formation (Figure 2a). After 200 cycles, the coated active material displayed half the cumulative irreversible capacity compared to the bare aggregated graphite particles, which results in electrochemical performances comparable to commonly used industrial battery-grade graphite.
This work highlights the clear advantages of X-ray nanotomography for precise assessment of the complex microstructure of battery electrodes down to the nanoscale. It demonstrates the impact of the application of electrode calendering for increasing energy density, and also establishes the benefits of petroleum pitch coating for increasing electrode cycle life and performance. Furthermore, it correlates irreversible lithium loss with the growth of the SEI layer in cycled electrodes, thus identifying a coating method to mitigate this loss. Overall, it shows the benefits of an innovative strategy for successful recycling and re-integration of waste graphite into the production chain of anodes for lithium-ion battery manufacturers, which could lead to increased productivity.
Principal publication and authors
From waste graphite fines to revalorized anode material for Li-ion batteries, J.C. Abrego-Martinez (a), Y. Wang (a), V. Vanpeene (b), L. Roué (a), Carbon 209, 118004 (2023); https://doi.org/10.1016/j.carbon.2023.118004
(a) Institut National de la Recherche Scientifique (INRS) - Centre Energie, Matériaux, Télécommunications (EMT), Varennes, QC, (Canada)
(b) ESRF
References
[1] P. Cloetens et al., Appl. Phys. Lett. 75(19), 2912-2914 (1999).
[2] G. Martínez-Criado et al., J. Synchrotron Radiat. 23(1), 344-352 (2016).
[3] B. Biber et al., Carbon 201, 847-855 (2023).
| About the beamline: ID16B |
|
ID16B is a hard X-ray nanoprobe dedicated to 2D or 3D analysis of nano-scaled materials combining X-ray fluorescence (XRF), diffraction (XRD), absorption spectroscopy (XAS), excited optical luminescence (XEOL), X-ray beam induced current (XBIC) and phase-contrast imaging. Low temperature, in-situ or operando sample environments can be accommodated. ID16B is dedicated to research areas with high scientific and societal impacts such as nanotechnology, earth and environmental sciences, and bio-medical research. |