- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2004
- Surface and Interface Science
- Atomic Structure of a Long-range Ordered Vicinal Surface of SrTiO3
Atomic Structure of a Long-range Ordered Vicinal Surface of SrTiO3
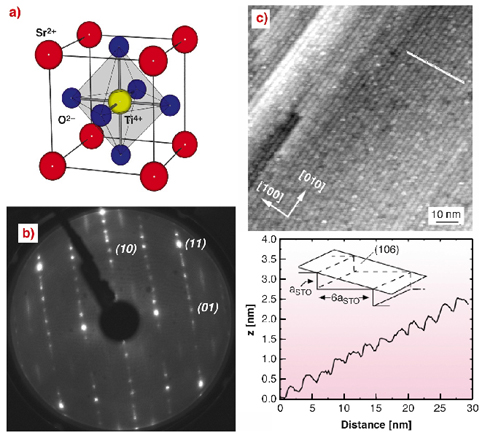
At the surface of a crystal, the atoms are missing some of their neighbours. For metals and semiconductors the atoms overcome this chemically frustrating situation by rearranging into a different short range order, i.e. the surfaces reconstructs. Reconstructions of semiconductors, for example, are mostly driven by rehybridisation and saturation of dangling bonds. Self organisation and long range order is initiated by the resulting stress. However, SrTiO 3 (STO, Figure 90a) is an insulator and characterised by strongly ionic bonds. The real charges of Sr, Ti, and O differ from the formal charges shown in Figure 90a and thus both (001) faces of STO (the SrO and TiO 2 low-index faces) are weakly polar [1], each carrying a charge of almost ± 1/2 e 0 per unit cell.
 |
|
Fig. 90: a) The 0.391 nm long STO unit cell; b) LEED image of the STO(106)-p(1x2) reconstruction. Extra lines and spots between [01] and [10] indicate a (1x2) reconstruction; c) Corresponding STM image (+1.5 V sample bias and 0.2 nA) and cross-section along the indicated line. |
The STO(106) surface, which is vicinal to STO(001), exhibits an unusual morphology. In the low energy electron diffraction (LEED) image of STO(106) in Figure 90b, besides the integer order reflections, spots at n/6 in the [10]-direction are due to the step structure and their sharpness is indicative of a very smooth (106) surface. The scanning tunnelling microscopy (STM) image in Figure 90c shows a very regularly "terraced" surface with an astonishing degree of long-range order. On the shorter length-scale, many uncorrelated contrast variations indicate substantial short-range disorder. The cross section of the STM image reveals rounded step edges.
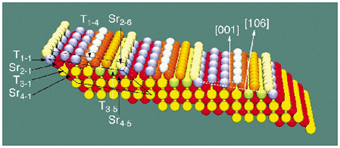
The structure of STO(106) was solved by diffraction measurements at beamline ID03 using X-rays of 20 keV energy. The surface was prepared in UHV by annealing only. Integrated intensities were recorded for 84 crystal truncation rods and the total number of non-equivalent reflections was 1644. From the widths of several reflections, the domain size was determined to be between 35 and 50 nm along the "step-edges", and close to 400 nm in the perpendicular direction, which represents a remarkable degree of long-range order. Utilising a "direct method", which has been developed for solving surface crystallographic problems [2], a starting model was obtained: a regularly stepped (106) surface with reduced site occupancies of the constituent atoms, varying as function of their proximity to the surface (Figure 91).
 |
|
Fig. 91: Resulting model of the STO(106) surface. The oxygen atoms, which are in bulk like positions, are not shown for clarity. Fully occupied Ti and Sr sites are represented by yellow and red colours, respectively. Lighter colours indicate lower occupancies. The continuous line defines the monoclinic surface cell used in the data analysis. |
The refinement of the surface structure yielded atomic coordinates very close to the corresponding bulk values and average occupancy values of the topmost Ti atoms (Ti1-1, Ti1-2, Ti1-3) of 0.5, compatible with a (disordered) p(1x2) surface cell, which explains the observed (1x2) LEED image (Figure 90b). The Ti and Sr population factors of deeper layers decrease gradually as a function of their atomic z-coordinates toward the surface. Figure 91 shows the resulting (106) surface with Ti and Sr atoms in an ordered p(1x2) surface cell. The Ti/O and the Sr/O- ratios of the two topmost surface layers are of 0.60 and 0.94, respectively, so both layers are less polar than in the bulk. The Ti/O ratio of the third surface layer is 0.47 and thus very close to the bulk value (0.5). The decrease in the site occupancy increases roughness/disorder on a short length scale, but renders the surface smoother on the long range.
The surface structure derived from the best fit exhibits STO(106) terraces, which are (001) planes, but with an alternating SrO1+![]() and TiO2-
and TiO2-![]() termination and thus alternating surface charges. This "charge undulation" represents the driving force behind the observed remarkable degree of long-range order. However, the adjustment of the cation/oxygen stoichiometry in conjunction with the varying cation site occupancy obviously decreases the "charge undulation" in order to further reduce the surface energy, rendering the surface more stable than exclusively SrO or TiO2 terminated STO(106) terraces.
termination and thus alternating surface charges. This "charge undulation" represents the driving force behind the observed remarkable degree of long-range order. However, the adjustment of the cation/oxygen stoichiometry in conjunction with the varying cation site occupancy obviously decreases the "charge undulation" in order to further reduce the surface energy, rendering the surface more stable than exclusively SrO or TiO2 terminated STO(106) terraces.
References
[1] J. Goniakowski, C. Noguera, Surf. Sci. 365, L657 (1996).
[2] see e.g.: D.K. Saldin, R.J. Harder, V.L. Shneerson, W. Moritz, Condens. Matter 13, 10689 (2001).
Principal Publications and Authors
X. Torrelles (a), J. Zegenhagen (b), J. Rius (a), T. Gloege (c), L.X. Cao (b) and W. Moritz (c), to be published.
(a) Institut de Ciència de Materials de Barcelona (C.S.I.C.), Bellaterra (Spain)
(b) ESRF
(c) Department für Geo- und Umweltwissenschaften, Universität München (Germany)



