- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2005
- Structural Biology
- Phasing of Macromolecular Structures using Radiation Damage
Phasing of Macromolecular Structures using Radiation Damage
The determination of novel macromolecular crystal structures is a critical step in the detailed understanding of many biological processes. During the course of structure determination, crystallographers must overcome the so called ‘phase problem’: while intensities of the X-rays diffracted by macromolecular crystals can be measured, phases cannot. The ESRF has a number of dedicated macromolecular crystallography (MX) beamlines where the energy of the incoming beam can be rapidly tuned to the absorption edge of the relevant heavy atom, thus enhancing the anomalous signal needed to solve the phase problem. These beamlines, together with fixed-energy MX stations as well as numerous other state-of-the-art and dedicated MX beamlines around the world, have made an extraordinary impact in structural biology.
Whereas these high intensity sources allow data collection at an unprecedented speed, they also painfully reveal the problem of radiation damage. Radiation damage quickly reduces the diffractive power of the cryocooled crystal, and can lead to specific changes within the structure. Heavy-atom substructures seem to be particular prone to damage, thus reducing anomalous signals, whereas small dispersive signals are easily swamped by non-isomorphism induced by the X-ray beam [1,2].
Rather than regarding radiation-damage as a problem to be avoided, we have shown that the specific nature of radiation-damage can actually be used to phase macromolecular structures [3]. The method has been named RIP; Radiation-damage Induced Phasing. It is tempting to compare the RIP method with the Single-Isomorphous Replacement (SIR) method, where two data sets are collected, one on a native crystal and one on a derivative. While collecting a highly redundant data set on a single crystal and splitting the data series into a number of complete, low-redundant data sets, the first data set could be seen as ‘the derivative’, whereas a later data set could be treated as ‘the native’. Existing software can then be used for solving the phase problem in a SIR-like manner.
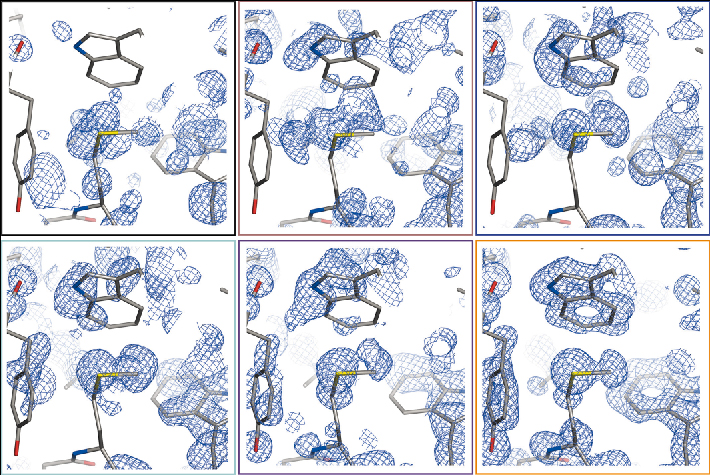
Six RIP data series have been collected on ID14-4 on crystals of native proteins: most structures could be phased solely using the radiation damage signal. We have shown that RIP may also be seen as a Multiple Isomorphous Replacement (MIR) experiment, where the native data set is missing. In contrast to MIR, a RIP substructure consists of a large number of small structural changes. Special software needs to be developed to take into account the presence of both gains (positive) and losses (negative) of electron density. We have demonstrated that careful scaling is needed to take the overall decay of the crystal into account. Existing packages have been adapted to exploit the specific nature of RIP: SHELXC and SHELXD for initial substructure determination while allowing the later data set to be ‘downscaled’, and SHELXE to allow substructure iteration whilst accounting for negative and positive electron densities (Figure 88).
 |
|
Fig. 88: Experimental RIP electron-density maps of lysozyme for different subsequent iterations in the structure determination using SHELXE. A total of 11 substructure iterations were made during which the substructure size increased from 8 to 40. Maps were drawn after iteration Nos. 1, 6, 8, 9, 10 and 11. |
Unlike SIR, MIR and MAD, RIP can be performed on crystals of native proteins. The technique is complementary to sulfur-SAD: the combined use of these techniques could become a “magic bullet” - overcoming the need to derivaterise proteins. The full maturation of the RIP technique requires the development of a new generation of software packages that can accurately model and exploit radiation damage. We have presented some initial steps towards this purpose.
References
[1] L.M. Rice, T.N. Earnest and A.T. Brunger, Acta Cryst. D56, 1413-1420 (2000).
[2] R.B.G. Ravelli, M.H. Nanao, A. Lovering, S. White and S. McSweeney, J. Synchrotron Rad. 12, 276-284 (2005).
[3] R.B.G. Ravelli, H.K. Leiros, B. Pan, M. Caffrey and S. McSweeney, Structure 11, 217-224 (2003).
Principal Publication and Authors
M.H. Nanao (a), G.M. Sheldrick (b) and R.B.G. Ravelli (a), Acta Cryst. D61, 1227-1237 (2005).
(a) EMBL, Grenoble Outstation (France)
(b) Georg-August Universität, Göttingen (Germany)



