- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2006
- Structural Biology
- Site specific recombination by the integron integrase: Broad DNA specificity
Site specific recombination by the integron integrase: Broad DNA specificity
Integron integrases catalyse recombination reactions between gene cassettes in bacteria, leading to the acquisition or dispersion of genetic traits. Enzymes in this group belong to the category of site specific recombinases from the tyrosine recombinase family. They possess a conserved set of active site residues (R-K-H-R-H/W-Y) which are involved in DNA cleavage, stabilisation of the phospho-tyrosine transition state intermediate and Holliday junction [1], and protonation of the leaving hydroxyl group. The catalysed reaction is isoenergetic with no ATP molecule required due to a concerted transfer of the high energy phospho-tyrosine bond to the phospho-diester DNA linkage. Site specific integrases use short stretches of double-stranded DNA which contain palindromic regions and a central asymmetric sequence as substrates. The integrons are mobile elements that carry an integrase enzyme, an insertion site (attI) and several gene cassettes composed of an open reading frame and an attC site. The attI site is around 40 base-pairs (bp) in length, whilst the attC site can vary from 40 bp to 160 bp, with a GTTRRRY (R:purine, Y:pyrimidine) consensus region. Integron integrases catalyse the integration of cassettes by recombining the attC sites with the attI, whilst they disperse cassettes by recombining attC sites. They have been shown to preferentially recombine the bottom strand of the recombination site.
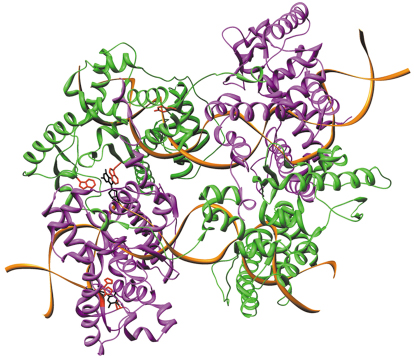
Structural information exists for the canonical enzymes namely Cre recombinase [2]. However, the structural basis of integron integrases use of varying sequences and single stranded DNA was still a conundrum. In order to address the mechanism of this reaction we proceeded with the determination of the three-dimensional structure of the integron integrase VchIntIa from Vibrio cholerae in complex with a target attC site reconstituted from a folded bottom strand (VCRbs), by X-ray crystallography. The structure was solved from a seleno-methionine derivatised protein-DNA crystal, using the single wavelength anomalous dispersion technique, with data recorded at beamline ID14-4. The crystal structure revealed a tetrameric arrangement of enzyme monomers on DNA, with two protein molecules bound to each DNA site. The VCRbs DNA includes key base-pair mismatches which were found to orient the enzyme on its substrate through specific recognition of the G20” base by a sandwich of conserved tryptophans (W157, W219), and of the T12” base by a stacking of histidines (H240, H241) and a proline (P232). The structure also explained the mechanism of regulation in the cleavage step whereby only a single cut is orchestrated in the complex due to the scaffolding of the ß4,5 hairpin in trans across the synapse thus upsetting the positioning of the tyrosine in the non-attacking subunit (Figure 63). This data and other recent findings are providing information on the role of enzymes that use base recognition without performing chemistry on the base itself. Site directed mutagenesis of the protein as well as the DNA bases of the VCRbs followed by biochemical electromobility shift assays or in vivo recombination frequency measurements agree with the structural information. We are now in the process of approaching the attI x attC complex by similar methods.
The ability of bacteria to recombine incoming gene cassettes based on structural recognition elements instead of sequence information per se greatly enhances their ability to adapt to changing environmental conditions. This study opens the field to finding more efficient ways of limiting antibiotic resistance acquisition and spread, as well as new molecular tools for manipulating DNA.
 |
|
Fig. 63: X-ray structure of the VchIntIa-VCRbs integron integrase–DNA complex. The synaptic complex is composed of a tetramer of enzyme molecules bound to two DNA hairpin stems. Specific recognition of extruded bases (G20” and T12”, black) by conserved residues (W157, W219 and H240-241, P232 respectively, red) orients the active site tyrosine Y302 (red), into attacking (green) and non-attacking (magenta) sub-units. Image copyright D. Gopaul/Institut Pasteur. |
References
[1] D.N. Gopaul, F. Guo, and G.D. Van Duyne, Embo J, 17(14), 4175-87 (1998).
[2] F. Guo, D.N. Gopaul, and G.D. van Duyne, Nature, 389(6646), 40-6 (1997).
Principal Publication and Authors
D. MacDonald, G. Demarre, M. Bouvier, D. Mazel and D.N. Gopaul, Nature 440, 1157-62 (2006).
Institut Pasteur, Paris (France)



