- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2006
- Structural Biology
- The crystal structures of the Dps1 and Dps2 from Deinococcus radiodurans reveal new metal binding sites
The crystal structures of the Dps1 and Dps2 from Deinococcus radiodurans reveal new metal binding sites
Deinococcus radiodurans (Dr) is an aerobic non-pathogenic bacterium able to withstand up to 10 kGy of ionising radiation whereas other organisms cannot survive a small fraction of this dose [1]. The Dr genome codes for two Dps (DNA protecting protein under starved conditions): DrDps1 and DrDps2 [1,2]. Their cellular functions are unknown but both proteins may function as iron storage proteins and have also been shown to bind DNA [2]. The combination of non-specific DNA binding with iron entrapment and detoxification is a strong beneficial combination of properties that the Dps proteins provide for the protection of cells. The determination of the DrDps structures, in the presence and absence of iron, sheds light on how these proteins function.
Like other members of the Dps family, DrDps1 and DrDps2 assemble as hollow spherical dodecamers of symmetry P23 with an outer shell diameter of around 90 Å and an interior diameter of 40 Å. Ferroxidase sites, formed between monomers confer fast iron oxidation capability to Dps, which in turn prevent the formation of highly cytotoxic reactive oxygen species (ROS) from the reaction of free Fe(II) with hydrogen peroxide or dioxygen [3,4]. In the DrDps2-Fe structure, a distinct iron site is observed 6.1 Å from the ferroxidase centre, probably representing a bio-mineralisation intermediate.
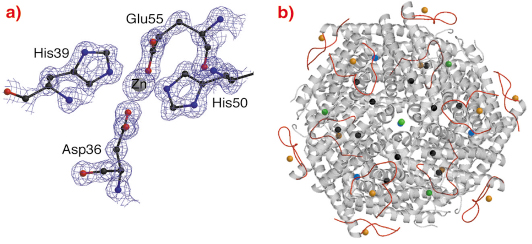
DrDps1 has a unique extended N-terminus where an unsuspected novel zinc binding site was identified (Figure 68). The N-terminal sequence also contains a number of positively charged residues and although the function of this sequence remains unclear, it is possible it functions as a metal sensor, or indeed interacts with DNA.
 |
|
Fig. 68: a) The DrDps1 Zn site at the external N-terminal extension of the protein dodecamer. The electron density map is contoured at 1.5 |
Two types of 3-fold related channels are thought to play a role in the transit of iron from the solvent to the ferroxidase centres. Clusters of negative electrostatic potential at the 3-fold N-terminal and C-terminal channels of DrDps1 guide cations to the target sites.
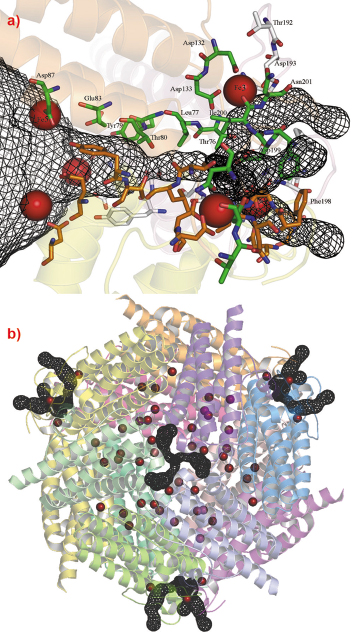
DrDps1 harbours a metal coordination site at the N-terminal side of the protein whereas in DrDps2 the equivalent metal binding site is found in a pocket located near to the external edge of the C-terminus (Figure 69). The latter is a non-specific metal site suspected to play a regulative role in iron uptake or release from the protein. In fact, the 3-fold C-terminal channel of DrDps2 can be thought of as an ion path able to control the flow of cations. The pathway is directed away from the centre of the 3-fold axis but passing through the metal site (Fe3, Figure 69). Under iron uptake conditions, iron can be captured at the external metallic site, followed by migration to the 3-fold axis channel and thereafter the centre of the protein. This is followed by iron capture by the ferroxidase centre and nucleation of the iron core.
 |
|
Fig. 69: a) The C-terminal pathway of DrDps2 with annotated residues forming the channel. The red spheres on the right are in the lateral channel pathway. b) The spherical assembly of DrDps2 showing iron locations. Each subunit is drawn in a different colour. The channel pathway in black isomesh is drawn with the plugin Caver for Pymol (www.pymol.org). |
Ongoing work focuses on the implications of metal binding to the non-specific site on the outer part of DrDps2 C-terminal channel, on the structural analysis of iron uptake and DNA interactions using small angle X-ray scattering and UV-visible spectroscopy.
References
[1] O. White, J.A. Eisen et al., Science 286(5444), 1571-7 (1999).
[2] A. Grove, and S.P. Wilkinson, J. Mol. Biol. 347(3), 495-508 (2005).
[3] K. Zeth, et al., Proc. Natl. Acad. Sci. U.S.A. 101(38), 13780-5 (2004).
[4] P. Wardman, and L.P. Candeias, Radiat. Res. 145(5), 523-31 (1996).
Principal Publication and Authors
M.G. Cuypers (a), E. Mitchell (a), S. McSweeney (a) et al., J. Mol. Biol. (in press); C. V. Romão (b) et al., J. Bio.l Inorg. Chem. 11: 891-902 (2006).
(a) ESRF
(b) ITQB, Oeiras (Portugal)



