Glasses and Ceramics
Mastering colouring effects in ceramics and glasses
In most cases, the esthetical value of glasses or ceramics result from optical effects (e.g. colour, opacity, or metallic shine). Such effects can be induced by the presence of specific compounds such as e.g. pigments, opacifying crystals, or metallic nanoparticles. The oxidation state of elements and more generally their chemical environment are directly correlated to these optical effects. Thus, historical glass and ceramic production methods required adequate control of firing conditions (temperature, atmosphere and time), as well as the introduction of oxidizing or reducing ingredients to master their final visual aspect. Colouring variations are usually obtained in ceramics and glasses by modulating the oxidation states of transition elements (such as Cr, Mn, Fe, Co, and Cu) due to their characteristic absorption frequencies in the visible region as a result of d–d electronic transitions. The energy range at ID21 (2.0–9.2 keV) is therefore very well suited to reveal the chemical state of these elements. XANES is an ideal technique to probe element speciation in amorphous glasses and/or in pigment micrometric grains.
|
|
M. Yuan and J. Bonet and H. Castillo-Michel and M. Cotte and N. Schibille and B. Gratuze and T. Pradell, "The role of tin and iron in the production of Spanish copper red glass from the 19th to the 20th century", Boletín de la Sociedad Española de Cerámica y Vidrio, (2023). |
Little is known about the materials used in the manufacture of red window glass in the 19th and the first decade of the 20th century. Here, we have studied fragments from eight Spanish glasses from the 19th and 20th centuries. The red glasses consist of a single layer of red glass on a colourless glass substrate. The chemical composition, oxidation state, nature of colourants and crystalline precipitates were determined by a selection of microanalytical techniques. In the study, we have found that the red colour of the glass is due to the presence of Cu0 nanoparticles, the red glass layer has lower calcium content than the substrate glasses, which we found favours copper being present in the glass predominantly in Cu+. They also contain tin and iron while lead, described in historical and modern documentation, was absent. Tin must have been added to the glass as a Sn2+ compound and acted as a reducing agent for copper. Iron is also a well-known reducing agent, but does not act as such in the red glasses studied here. However, it may have facilitated the reduction of copper to Cu0 by promoting its incorporation into the glass as Cu+ rather than Cu2+.
|
Mingyue Yuan and Jordi Bonet and Marine Cotte and Nadine Schibille and Bernard Gratuze and Trinitat Pradell, "The role of sulphur in the early production of copper red stained glass", Microscopy and Microanalysis, 1-10 (2023) |
|
Little is known about the production of ruby red copper stained glasses from the Medieval and Renaissance periods apart from the fact that the colour is due to the presence of small metallic copper nanoparticles and that tin, the most common reducing agent used in copper red glass production since the 19th century, is not present. In fact, very few workshops in Europe were able to make red glass in historical times, and they kept it secret, so very little is known about how it was obtained. These workshops exported the red glass throughout Europe. Recently, the presence of copper sulphide particles and the data obtained in the replication red glass following historical recipes suggested that sulphur might be the key ingredient in this process.
Here, a collection of historical red glasses from these periods has been analysed using a combination of microanalytical techniques; Electron Microprobe (EM) and Field Emission and Scanning Electron Microscopy (FESEM) to verify the chemical composition and nanostructure of the glasses, Synchrotron radiation micro-X-Ray Diffraction (micro-XRD) to establish the nature of the nanocrystalline precipitates, and S, Cu and Fe K-edge micro-X-Ray Absorption Spectroscopy (micro-XAS) to determine the speciation. The data obtained show that the oxidation of S2− into S6+ in the glass is responsible for the precipitation of copper nanoparticles. The development of a sulphide-silicate partition and the presence of Fe3+ in the melt give rise to the precipitation of the high-pressure tetragonal polymorph of chalcocite (Cu2S). Differences between the Medieval and Renaissance red glass are determined.
 |
L. Verger, O. Dargaud, G. Rousse, M. Cotte and L. Cormier, "The stability of gahnite doped with chromium pigments in glazes from the French manufacture of Sèvres", Journal of the American Ceramic Society, 100, 86-95 (2017). |
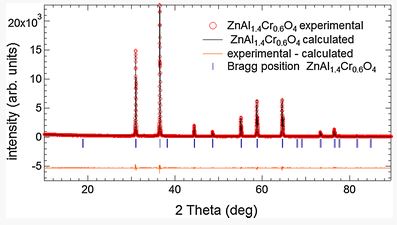
The French manufacture of Sèvres, famous for its production of fine porcelain artifacts, has been synthesizing pink pigments mainly composed of the spinel phase ZnAl2−xCrxO4 with x varying from 0.25 to 0.41 since the middle of the 19th century. This kind of pigment is mixed with an uncolored frit to obtain decorations for porcelain artifacts. However, the pink color of the pigment is altered in a particular uncolored frit and a brownish color appears. The mechanism of this color change was investigated. Observations under a scanning electron microscope revealed the formation of a phase rich in Cr resulting from reactions between the uncolored frit and the pigment during firing. X‐ray diffraction combined with Rietveld refinements and X‐ray absorption near edge structure measurements at the Cr K‐edge showed that the new formed phase belongs to the same spinel phase ZnAl2−xCrxO4 than the pigment, but with a higher Cr content x. We showed that its formation and thus the stability of the pigment is driven by the Al content in the uncolored frit.
 |
L. Verger, O. Dargaud, G. Rousse, E. Rozsályi, A. Juhin, D. Cabaret, M. Cotte, P. Glatzel and L. Cormier, "Spectroscopic properties of Cr 3 + in the spinel solid solution ZnAl2−xCrxO4", Phys Chem Minerals, 43, 33-42 (2015). |
The evolution of the structural environment of Cr3+ along the solid solution ZnAl2−xCrxO4 has been investigated using a multi-analytical approach. X-ray diffraction confirms that the system follows Vegard’s law. Diffuse reflectance spectra show a decrease of the crystal field parameter with the Cr content, usually related to the increase of the Cr–O bond length in a point charge model. This interpretation is discussed and compared to the data obtained by first principle calculations based on density functional theory. X-ray absorption near edge structure spectra at the Cr K-edge show a pronounced evolution in the pre-edge with the Cr content, characterised by the appearance of a third feature. Calculations enable to assign the origin of this feature to Cr neighbours. The colour change from pink to brownish pink and eventually green along the solid solution has also been quantified by calculating the L*, a*, b* and x, y coefficients in the system defined by the International Commission on Illumination.
T. Wang, T. Q. Zhu, Z. Y. Feng, B. Fayard, E. Pouyet, M. Cotte, W. De Nolf, M. Salomé and P. Sciau, "Synchrotron radiation-based multi-analytical approach for studying underglaze color: The microstructure of Chinese Qinghua blue decors (Ming dynasty)", Analytica Chimica Acta, 928, 20-31 (2016). |
|
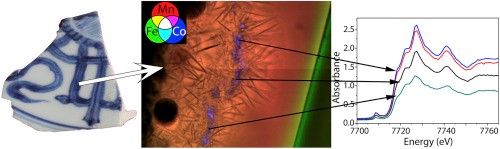
In this paper, we develop a methodological approach combining macro-X-ray fluorescence and synchrotron radiation-based techniques (μXRF, full-field XANES and μXRD) to determine the composition and microstructure of underglaze decors of Qinghua porcelains (Ming dynasty). Various transition metal elements (Fe, Mn, Co) are present in the blue decoration of these ceramics and the approach proposed allows for establishing the feature of each. Thus it shows that Fe ions are distributed homogeneously over the whole glaze without any significant difference in blue and white parts. They do not play a significant role in the color. In contrast, Co ions exhibit a heterogeneous distribution with CoAl2O4 particles close to the body/glaze interphase. These particles play a key role in the blue color and, the hue variations seem in greater part to link to their density and repartition. Co dispersed in the glassy matrix is also bivalent and mainly in tetragonal coordination, leading also to a blue color. Mn ion distribution is similar to the one of Co but without presenting local high concentrations associated to Mn based particles. Mn affects the darkness of the color and for the sample without CoAl2O4 particle; it is the main color contribution. The presence of CoAl2O4 crystals was confirmed by μXRD, which revealed, in addition, a variation of cell parameters certainly linking to a Co partial substitution.
 |
Cianchetta, K. Trentelman, J. Maish, D. Saunders, B. Foran, M. Walton, P. Sciau, T. Wang, E. Pouyet, M. Cotte, F. Meirer, P. A. Pianetta and A. Mehta, "Evidence for an Unorthodox Firing Sequence Employed by the Berlin Painter: Deciphering Ancient Ceramic Firing Conditions through High-Resolution Material Characterization and Replication", Journal of Analytical Atomic Spectrometry, 30, 666-676 (2015). |
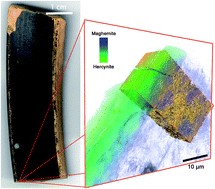
XANES spectroscopy was used to complement the results previously obtained with Raman spectroscopy by the same group to determine the firing conditions used in the production of a single vessel painted by the Berlin Painter in the 5th century B.C. The vessel, part of the collection of the J. Paul Getty Museum, presents a complicated layered architecture of black and red gloss, with different stratigraphies present on the interior and exterior surfaces. The study of two samples, one each from the interior and exterior surface of the vessel, was performed with the complementary analytical techniques of X-ray nano- and micro-spectroscopy (X-ray fluorescence spectroscopy (XRF) and full-field transmission X-ray micro-spectroscopy (FF-XANES) across the Fe K edge), and supported by a replication study. The replicates, made in a laboratory furnace providing complete control over the firing temperature and oxygen partial pressure, provided a paradigm for the comparison of the mineralogical phases observed in the ancient samples, which led to a deeper understanding of the firing conditions necessary for the production of the Berlin Painter's vessel. Our results confirm the necessity of multiple firings and painting applications to obtain the Berlin Painter's architecture and provide a further example of the multiplicity of techniques and practices employed by the potters of the Kerameikos in ancient Athens.
 |
F. Meirer, Y. Liu, E. Pouyet, B. Fayard, M. Cotte, C. Sanchez, J. C. Andrews, A. Mehta and P. Sciau, "Full-field XANES analysis of Roman ceramics to estimate firing conditions-A novel probe to study hierarchical heterogeneous materials", Journal of Analytical Atomic Spectrometry, 28 1870 - 1883 (2013). |
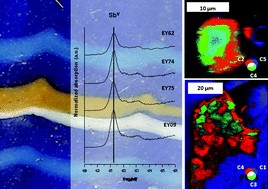
Roman black gloss ceramics from two different locations and separated by 50–80 years were investigated by X-ray absorption near edge structure analysis in full field hard X-ray transmission microscopes. These spectro-microscopy measurements were complemented by Raman spectroscopy, and X-ray diffraction spot analyses to gain insights into possible differences in manufacturing technology. Our results indicate that the two vessels underwent significantly different firing protocols, suggesting that there was a surprisingly quick evolution of a complex technological process in response to changing needs and tastes of a burgeoning empire. Furthermore, our results show that the ability of the full field X-ray spectro-microscope to investigate large sample areas (from hundreds of µm2 to as much as 2 mm2) with high spatial resolution (of 300 nm down to 30 nm) together with its ability to correlate sample porosity (derived from tomography) with the distribution of chemical phases makes it an invaluable tool in the investigation of nanoscale processes in hierarchically heterogeneous chemical systems—from Roman ceramics to some of the most advanced technological products of today.
 |
S. Lahlil, M. Cotte, I. Biron, J. Szlachetko, N. Menguy and J. Susini, "Synthesizing lead antimonate in ancient and modern opaque glass", Journal of Analytical Atomic Spectrometry, 26, 1040-1050 (2011). |
Through the study of lead antimonate based opacifiers in three opaque glass productions—Egyptian glass of the 18th dynasty (1570–1292 BC), Roman mosaic tesserae and beads from Aquilea and Rome (2nd c. BC–5th c. AD) and Nevers lampworking glass figures 18th c. AD)—this paper shows the evolution of lead antimonate production during different periods of History. We also show the necessity of using systematic micro-chemical analyses, with both high spatial and high energy resolution techniques to investigate these types of materials. The synchrotron-based μ-XANES measurements combined with the microstructure observations (SEM and TEM), the chemical and structural analyses (EDX, WDS, μ-Raman), is the first step to getting information on the raw materials used and the technological processes employed to produce lead antimonate. The heterogeneity from one sample to another but also within the same sample, and even further within a single crystal aggregate clearly shows that a production cannot be unambiguously associated to a single chemical composition. However, differences between the three productions are clearly highlighted and hypotheses about glass manufacturing are proposed.
 |
S. Lahlil, I. Biron, M. Cotte and J. Susini, "New insight on the in situ crystallization of calcium antimonate opacified glass during the Roman period", Applied Physics A, 100, 683-692 (2010). |
Glass is usually opacified by small crystalline particles, called opacifiers, dispersed in the translucent vitreous matrix. To understand the glassmaking conditions used to produce calcium antimonate opacified glass, the in situ crystallization process has been studied through synthetic glasses made in the laboratory. The effects of the nature and the concentration of the antimony source, the temperature and the duration of heat-treatments on the calcium antimonates crystallization have been tested. The physico-chemical characteristics of these glasses were compared to Roman mosaic tesserae from Aquilea and Rome (1st cent. B.C.–6th cent. A.D.). We show that the glass composition (EDX), the microstructure (SEM-BSE, imaging treatment), the oxidation state of antimony in the vitreous matrices (μ-XANES) and the proportion of the crystalline phases (XRD with Rietveld refinement) are suitable parameters to assess glassmaking conditions used in ancient times. We demonstrate that opaque Roman glasses were obtained by in situ crystallization, probably using roasted stibnite Sb2O4 and by doing heat-treatment between 1 or 2 days.
 |
S. Lahlil, I. Biron, M. Cotte, J. Susini and N. Menguy, "Synthesis of calcium antimonate nano-crystals by the 18th dynasty Egyptian glassmakers", Appl. Phys. A, 98, 1-8 (2010). |
During the 18th Egyptian dynasty (1570–1292 B.C.), opaque white, blue and turquoise glasses were opacified by calcium antimonate crystals dispersed in a vitreous matrix. The technological processes as well as the antimony sources used to manufacture these crystals remain unknown. Our results shed a new light on glassmaking history: contrary to what was thought, we demonstrate that Egyptian glassmakers did not use in situ crystallization but first synthesized calcium antimonate opacifiers, which do not exist in nature, and then added them to a glass. Furthermore, using transmission electron microscopy (TEM) for the first time in the study of Egyptian opaque glasses, we show that these opacifiers were nano-crystals. Prior to this research, such a process for glassmaking has not been suggested for any kind of ancient opaque glass production. Studying various preparation methods for calcium antimonate, we propose that Egyptian craftsmen could have produced Ca2Sb2O7 by using mixtures of Sb2O3 or Sb2O5 with calcium carbonates (atomic ratio Sb/Ca=1) heat treated between 1000 and 1100°C. We developed an original strategy focused on the investigation of the crystals and the vitreous matrices using an appropriate suite of high-sensitivity and high-resolution micro- and nano-analytical techniques (scanning electron microscopy (SEM), X-ray diffraction (XRD), TEM). Synchrotron-based micro X-ray absorption near edge spectroscopy (μ-XANES) proved to be very well suited to the selective measure of the antimony oxidation state in the vitreous matrix. This work is the starting point for a complete reassessment not only of ancient Egyptian glass studies but more generally of high-temperature technologies used throughout antiquity.
 |
R. Arletti, G. Vezzalini, S. Quartieri, D. Ferrari, M. Merlini and M. Cotte, "Polychrome glass from Etruscan sites: First non-destructive characterization with synchrotron µXRF, µXANES and XRPD", Applied Physics A, 92, 127-135 (2008). |
This work is devoted to the characterization of a suite of very rare, highly decorated and coloured glass vessels and beads from the VII to the IV century BC. The most serious difficulty in developing this study was that any sampling – even micro-sampling – was absolutely forbidden. As a consequence, the mineralogical and chemical nature of chromophores and opacifiers present in these Iron Age finds were identified by means of the following synchrotron-based, strictly non-destructive, techniques: micro X-ray fluorescence (μ-XRF), Fe K-edge micro X-ray absorption near edge spectroscopy (μ-XANES) and X-ray powder diffraction (XRPD). The μ-XRF mapping evidenced high levels of Pb and Sb in the yellow decorations and the presence of only Sb in the white and light-blue ones. Purple and black glass show high amounts of Mn and Fe, respectively. The XRPD analyses confirmed the presence of lead and calcium antimonates in yellow, turquoise and white decorations. Fe K-edge μ-XANES spectra were collected in different coloured parts of the finds, thus enabling the mapping of the oxidation state of these elements across the samples. In most of the samples iron is present in the reduced form Fe2+ in the bulk glass of the vessels, and in the oxidized form Fe3+ in the decorations, indicating that these glass artefacts were produced in at least two distinct processing steps under different furnace conditions.





