- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Structural biology
- X-ray snapshots of likely intermediates in the synthesis and degradation of biological Fe4S4 clusters
X-ray snapshots of likely intermediates in the synthesis and degradation of biological Fe4S4 clusters
Fe-S clusters are ancient protein-bound prosthetic groups generally involved in substrate binding and activation, electron transfer and stress sensing in the regulation of gene expression. Alternatively, they can also play a structural role. Their compositions vary from the commonly found Fe2S2, Fe3S4 and Fe4S4 centres to the more complex Fe7S9Mo cluster found in the active site of nitrogenase. Facultative microbes can take advantage of the intrinsic sensitivity of Fe-S clusters to O2-induced damage to switch between anaerobic and aerobic metabolisms, a property used by the fumarate nitrate reduction regulator (FNR). Under 0.5% oxygen, FNR is a protein dimer that coordinates one Fe4S4 cluster per monomer and binds specific DNA sequences to either activate or repress gene expression. Conversely, under higher oxygen levels, there is a rapid Fe4S4-to-Fe2S2 cluster transformation upon which FNR dissociates into monomers that no longer bind DNA. The three dimensional structure of FNR remains unsolved, which imposes a severe limitation in the understanding of this process.
It was thought that Fe4S4-to-Fe2S2 cluster degradation in FNR involved the release of one Fe2+, one Fe3+ and two S2- ions [1]. However, a very recent study, which combined resonance Raman (RR), UV-visible absorption/CD spectroscopy and mass spectrometry [2], indicates that FNR cluster conversion results in an Fe2S2 cluster that retains two sulfides as cysteine persulfide ligands. The relevant reactions are:
Fe4S42+ + O2 → Fe3S4+ + Fe2+ + O2•– (1)
2 Cys-S– + Fe3S4+ + O2 + 4 H+ → 2 Cys-S-S- + Fe2S22+ + Fe3+ + 2 H2O (2)
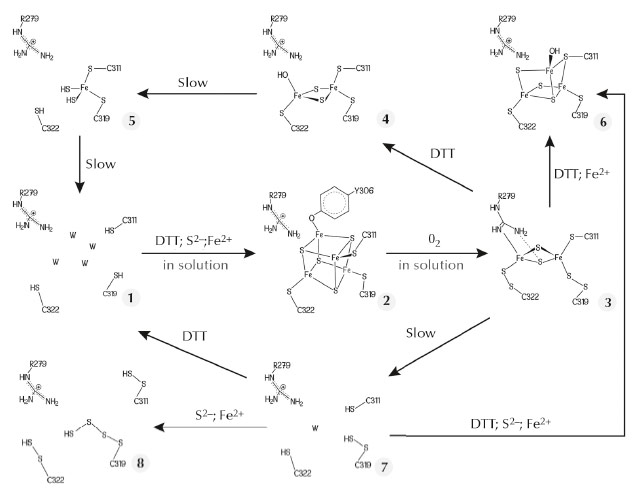
The FeFe-hydrogenase maturase HydE from Thermotoga maritima, which belongs to the radical S-adenosyl-L-methionine (SAM) protein super-family, binds the Fe4S4 cluster typical of these enzymes, along with a more exposed centre that displays variable compositions in our crystals. We have used this exposed site as a template to obtain unprecedented Fe-S clusters that most likely constitute a sequence describing cluster degradation and assembly (Figure 48). The cluster with the highest number of Fe ions in our original crystals resembles a standard Fe3S4 centre, except that one cysteine ligand bridges two iron ions (6 in Figure 48). We subsequently obtained a new crystal form of TmHydE using a higher salt concentration [3]. This form contains a regular Fe4S4 cluster, as previously reported for this protein in solution [4], that has a tyrosine side chain as its fourth protein ligand (2 in Figure 48 and Figure 49). Most relevant to the FNR spectroscopic results [2] is our finding of a structure where the Fe2S2 cluster is coordinated by two cysteine persulfide ligands (3 in Figure 48). Persulfide formation must have resulted from sample exposure to traces of O2 either in the glove box or during its transfer to it.
 |
|
Fig. 48: Postulated Fe-S cluster degradation pathway based on our different TmHydE crystal structures. Unless specified, all reactions were performed within crystals. W in species 1 and 7 represents water molecules. |
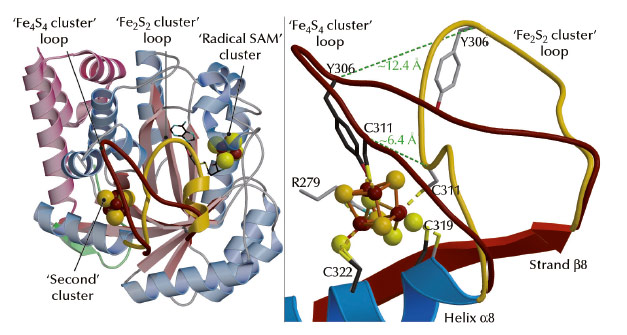
A comparison between our structures shows that the reversible transition from the Fe4S4 cube to the Fe2S2 rhomb requires a major rearrangement of the coordination site in TmHydE (Figure 49). Comparable changes may be responsible for the O2-induced dimer dissociation in FNR [5]. Taken together, our results and the RR spectroscopic studies mentioned above [2] provide a coherent functional and structural description of the Fe4S4-to-Fe2S2 cluster conversion that generally takes place upon O2 exposure in Fe-S cluster-containing proteins. The main proposition derived from our work [3] and reference [2] is the existence of a Fe-S cluster direct repair mechanism involving the reduction of persulfide ligands in a Fe2S2 cluster similar to the transition that took place in our crystals:
2 Cys-S-S- + Fe2S22+ + 2 Fe2+ + 4 e- → 2 Cys-S- + Fe4S42+(3)
Such a mechanism represents a considerable economy to the cell because only Fe ions need to be available for the reconstitution of Fe4S4 clusters in FNR under anaerobic conditions.
 |
|
Fig. 49: Structural differences between the Fe4S4 and Fe2S2 cluster-containing TmHydE proteins. (Left) View of the overall structure depicting the movement of a loop induced by cluster conversion (shown in brown for Fe4S4 and gold for Fe2S2). (Right) Close-up view of the Fe-S cluster-binding site (colour codes as in left figure). Dashed lines indicate the shifts of positions of the Ca atoms of C311 and Y306 when the two structures are compared. |
Principal publication and authors
Y. Nicolet, R. Rohac, L. Martin and J.C. Fontecilla-Camps, PNAS 110, 7188–7192 (2013).
Metalloproteins Unit, Institut de Biologie Structurale J.-P. Ebel CEA-CNRS-UJF, Grenoble (France)
References
[1] J.C. Crack, A.A. Gaskell, J. Green, M.R. Cheesmant, N.E. Le Brun and A.J. Thomson, J. Am. Chem. Soc. 130, 1749-58 (2008).
[2] B. Zhang, J.C. Crack, S. Subramanian, J. Green, A.J. Thomson, N.E. Le Brun, et al., Proc Natl Acad Sci USA 109, 15734-9 (2012).
[3] Y. Nicolet, R. Rohac, L. Martin and J.C. Fontecilla-Camps, Proc Nat Acad Sci USA 110, 7188-92 (2013).
[4] J.K. Rubach, X. Brazzolotto, J. Gaillard and M. Fontecave, FEBS Lett. 579, 5055-60 (2005).
[5] B.A. Lazazzera, H. Beinert, N. Khoroshilova, M.C. Kennedy and P.J. Kiley, J. Biol. Chem. 271, 2762-8 (1996).



