- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2014
- Dynamics and extreme conditions
- Diverse chemistry in the magnesium/carbon system
Diverse chemistry in the magnesium/carbon system
Magnesium and carbon, both of which form numerous compounds with other elements, have a remarkably low affinity for one another. At ambient pressure, only the reaction of Mg or MgO with hydrocarbons leads to the formation of metastable carbides with reasonable yields, and pure Mg and carbon do not react to form stable compounds at any temperature. With experimental data obtained at beamline ID06LVP, we have demonstrated that thermodynamically stable Mg-C compounds are indeed possible under high-pressure conditions (above 5 GPa and 1300 K). The new compound synthesised, β-Mg2C3, crystallises in the monoclinic C2/m space group, and contains rare allene-derived C34- anions that are isoelectronic with CO2.
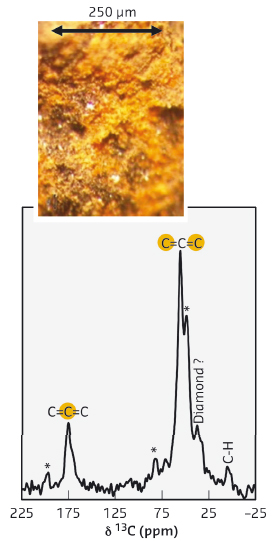
A bright golden-yellow powder of this new Mg-C compound was recovered from high-pressure, high-temperature experiments (Figure 81). Mass spectrometry analysis of the hydrolysis products showed allene, C3H4, indicative of the presence of C34- anions within the structure. In addition, 13C NMR analysis confirmed the presence of two structurally distinct carbon atoms in an approximate 1:2 ratio, and Raman spectra indicated carbon stretching modes from [C=C=C]. The crystal structure –clearly distinct from previously reported compounds– was, however, impossible to solve using the available powder X-ray diffraction data.
 |
|
Fig. 81: 13C MAS NMR spectrum of β-Mg2C3 recovered from 9 GPa showing two unique carbon environments. Inset: optical microscope picture of the golden-yellow powder. |
To resolve the structure of this new magnesium carbide, two important steps were taken. First, potential Mg2C3 structures were predicted using USPEX, an ab initio structural evolution algorithm. Second, in situ high-pressure synthesis was performed at beamline ID06 using the recently-commissioned large-volume press. The in situ synthesis allowed the nearly phase-pure synthesis of the new compound, and high-resolution X-ray diffraction patterns obtained allowed conclusive comparison with theoretical models.
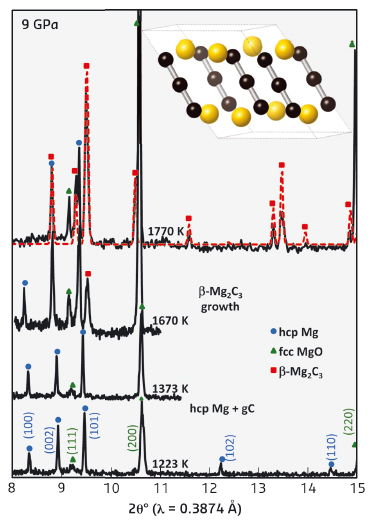
Remarkably, the high-pressure Mg2C3 structure predicted via USPEX was a perfect match to the experimental X-ray diffraction data (Figure 82). Thus, the structure of β-Mg2C3 was solved. Unlike α-Mg2C3, which contains alternating layers of C34- chains oriented in opposite directions, all C34- chains within β-Mg2C3 are nearly aligned along the crystallographic c-axis.
 |
|
Fig. 82: In situ diffraction data collected during HPHT synthesis at ID06. Crystal structure of β-Mg2C3 showing C34- ions aligned along the c-axis. |
After exploring pressure as an additional dimension for the chemistry of the Mg−C system, four magnesium carbides are now known: (1) tetragonal MgC2, (2) orthorhombic α-Mg2C3, (3) monoclinic β-Mg2C3, and (4) cubic Mg2C [1]. Taking into account that, at ambient pressure and at pressures up to ~5 GPa, the elements do not interact at any temperature, it is quite astonishing to observe such rich chemistry.
Finally, these results indicate that the Mg-C system should be completely revised under high pressure [2]. The phase behaviour in this system is important for the production of structurally-perfect diamonds with high growth rates as compared to other known metal-carbon systems, and also for the synthesis of unique nitrogen-free and boron-doped semiconductive diamonds. With these new results, we now have a much deeper understanding of Mg-C thermodynamics under extreme conditions.
Principal publication and authors
T.A. Strobel (a), O.O. Kurakevych (a, b), D.Y. Kim (a), Y. Le Godec (b), W. Crichton (c), J. Guignard (c), N. Guignot (d), G.D. Cody (a) and A.R. Oganov (e-h), Inorg. Chem. 4, 53, 7020−7027 (2014).
(a) Geophysical Laboratory, Carnegie Institution of Washington, Washington DC (USA)
(b) Institut de Minéralogie, de Physique des Matériaux et de Cosmochimie, UPMC,CNRS, Paris (France)
(c) ESRF
(d) SOLEIL, Gif-sur-Yvette (France)
(e) Department of Geosciences, SUNY, Stony Brook, NY (USA)
(f) Institute for Advanced Computational Science, SUNY, Stony Brook, NY (USA)
(g) Moscow Institute of Physics and Technology, Dolgoprudny City (Russian Federation)
(h) Northwestern Polytechnical University, Xi’an (China)
References
[1] O.O. Kurakevych, T.A. Strobel, D.Y. Kim and G.D. Cody, Angew. Chem. Int. Ed. 52, 8930-8933 (2013).
[2] O.O. Kurakevych, Y. Le Godec, T.A. Strobel, D.Y. Kim, W.A. Crichton and J. Guignard, J. Phys. Chem. C 118, 8128–8133 (2014).



