- Home
- News
- General News
- X-ray unveils the...
X-ray unveils the creation process of materials on several length scales
20-07-2021
Nanostructuring often makes materials very powerful in many applications. Some nanomaterials take on the desired complex structures independently during their creation process. Scientists from the University of Hamburg, DESY, ESRF and the Ludwig Maximilians University in Munich have studied the formation of cobalt oxide crystals just a few nanometers in size and how they assemble, while they are still being formed. The results are published today in Nature Communications.
Nanomaterials have special properties that make them more effective than conventional materials in various applications. In sensors and catalysts (in green energy production, such as water splitting into energy-rich hydrogen and oxygen) the important chemical processes happen at the surface. Nanostructured materials, even in small amounts, provide a very large surface and are therefore suitable for this kind of applications.
Further potential arises due to the variety of shapes and material combinations that are conceivable on the nanoscale. However, establishing the exact shape of these nanostructures can be a tedious process. Researchers focus on nanocrystals that independently form complex structures without any external influence, for example by sticking together (assembling). This increases their effectiveness in important technological applications, such as green energy generation or sensor technology.
“Often nanoparticles arrange themselves independently, as if following a blueprint, and take on new shapes,” explains Lukas Grote, one of the main authors of the study and scientist at DESY and the University of Hamburg. “Now, however, we want to understand why they are doing this and what steps they go through on the way to their final form. That is why we follow the formation of nanomaterials in real time using high-intensity X-rays. ” For some of the experiments, the researchers used the European Synchrotron Radiation Facility (ESRF) and DESY's synchrotron radiation source PETRA III.
However, nanomaterials usually have a complex formation genesis, emphasizes Dorota Koziej, professor at the Center for Hybrid Nanostructures (CHyN) at the University of Hamburg: “That is why a single X-ray method is not sufficient to examine all the steps during the emergence of nanostructures. At every step you have to apply the right technique and later put the individual results together to form an overall picture. "
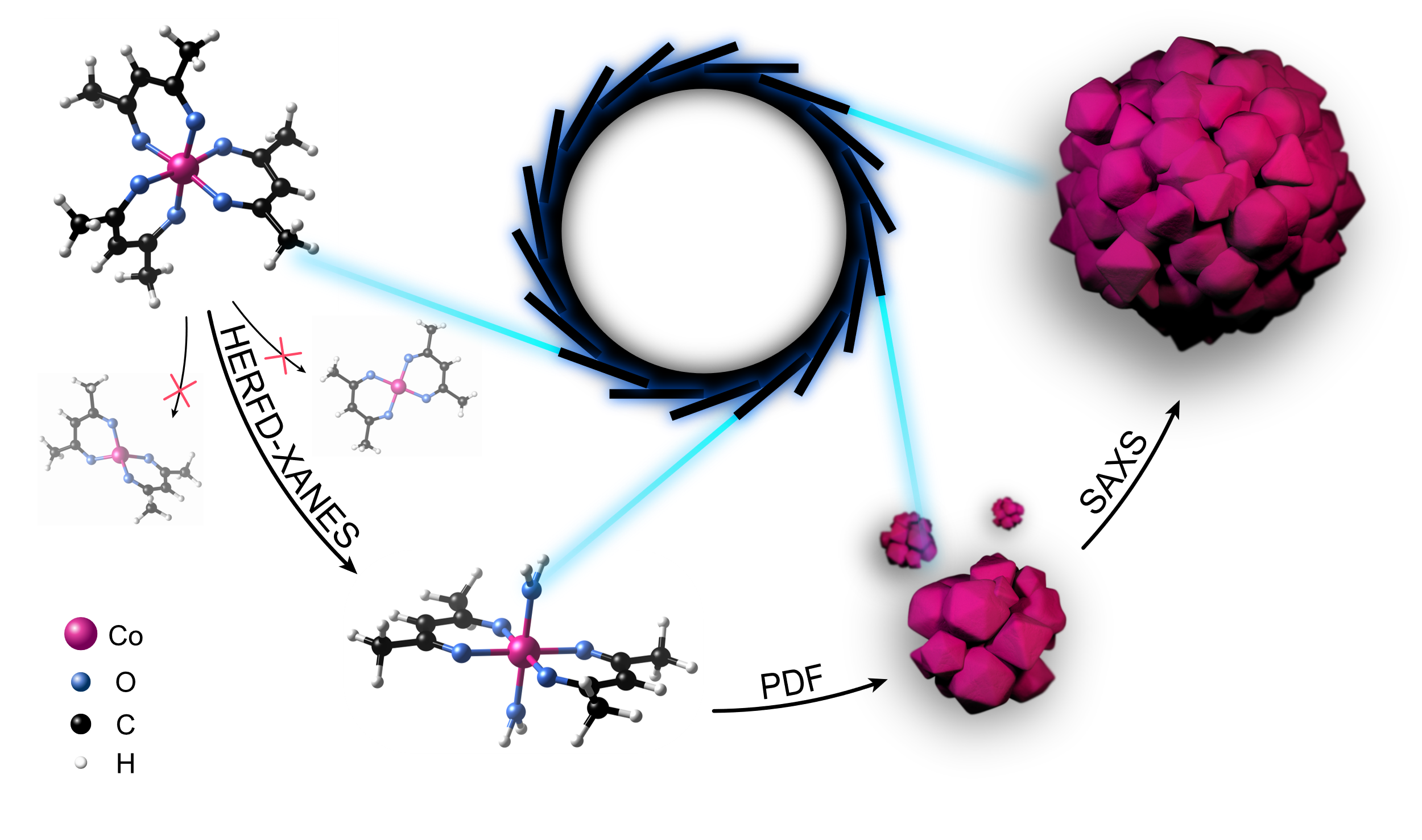 |
|
X-rays from a synchrotron radiation source are both attenuated (absorbed) and deflected (scattered) by matter. Depending on which of these interactions is measured with a certain X-ray technology, conclusions can be drawn about different stages of the development process of a nanomaterial. If you combine both X-ray absorption and X-ray scattering, you can decipher all the steps from the starting material (left) to the fully assembled nanostructures (right). Credits: Nature Communications. |
When X-rays move through matter, they are both attenuated (absorbed) and changed (scattered) in their direction of movement. Both processes can be observed separately, and conclusions can be drawn from them about different stages in the growth process of a nanomaterial. From the X-ray absorption spectroscopy, chemical changes in molecules can be recognised. This technique shows the steps in which the starting materials of the nanostructures are transformed, even before nanocrystals have formed.
At the ESRF, the team carried out high resolution X-ray spectroscopy experiments on ID26 and BM14. Blanka Detlefs, co-author of the paper and scientist on ID26, explains: “our technique allows to follow chemical state changes in complex reactions within a chemical reactor and in real time. Such in situ experiments are always challenging, as we have to reproduce the same experimental conditions as in the laboratory with an extra optical access for the X-ray beam.” Kristina Kvashnina, researcher at the Helmholtz-Zentrum Dresden Rossendorf and also involved in the experiment, adds: “Combining the technique with electronic structure calculations, it becomes an efficient method to study and evaluate the unique properties of nanomaterials”.
From the moment when the smallest nanocrystals form and grow, the way in which the X-ray light is scattered by the material changes. “The superposition (interference) of light waves causes more light to be deflected in certain directions than in others. From this scattering of the X-ray light, we can then calculate how the shape and size of the cobalt oxide nanocrystals develop during their formation process,” explains Cecilia Zito, one of the main authors, from the University of Hamburg and who is now working at the Sao Paulo State University in Brazil. “We can then also observe the way in which the nanocrystals attach to each other, that is, assemble independently to form new and more complex structures,” adds Kilian Frank, also main author from the Ludwig Maximilians University in Munich.
“Finding out the blueprint for nanomaterials still remains a major challenge. Every compound is different, and we don't yet understand many ways of self-assembly,” says Grote. However, the researchers involved are convinced that this will soon change. "In the end, we are looking for an overall model with which we can explain and predict the formation of complex nanostructures in general," says Koziej. "In this way we learn how to best influence the dynamic processes on the nanoscale, and ultimately how to use the full potential of these tailor-made nanomaterials."
Reference:
Grote L. et al, Nature Communications, DOI: 10.1038/ s41467-021-24557-z CC



