- Home
- News
- Spotlight on Science
- Diversifying nucleoside...
Diversifying nucleoside analogues with promiscuous phosphorylases
18-11-2021
Diffraction data collected at beamline ID29 shine light on the activity of promiscuous thermostable nucleoside phosphorylases. New research shows how these enzymes can be used to diversify nucleoside analogues, granting access to potential new antiviral and anticancer compounds.
Enzymes are empowering tools in organic chemistry. However, shaped by evolution, their natural substrate scopes are often more limited than desired by many chemists. X-ray diffraction data collected at beamline ID29 show how minor active site modifications in pyrimidine nucleoside phosphorylases expand their substrate scope to include nucleoside analogues carrying alkylated sugars. These promiscuous enzymes enable straightforward and selective access to a range of modified nucleosides from a single precursor - a transformation which is impossible to achieve with conventional synthetic chemistry [1].
Nucleosides are important biomolecules. All life on earth employs nucleosides as enzymatic cofactors, building blocks of DNA and RNA or as energy transport systems. Consequently, nucleoside analogues, which mimic their natural counterparts but disturb or alter certain cellular functions, have been attracting the attention of medicinal chemists for several decades. Today, nucleoside analogues are ubiquitous in the life sciences. They serve as pharmaceuticals for the treatment of various cancers and viral infections, and have become indispensable as molecular biology tools. For instance, the anti-HIV drug islatravir or the anti-COVID-19 drug candidate Molnupiravir are both nucleoside analogues carrying subtle but crucial modifications compared to their parent compounds.
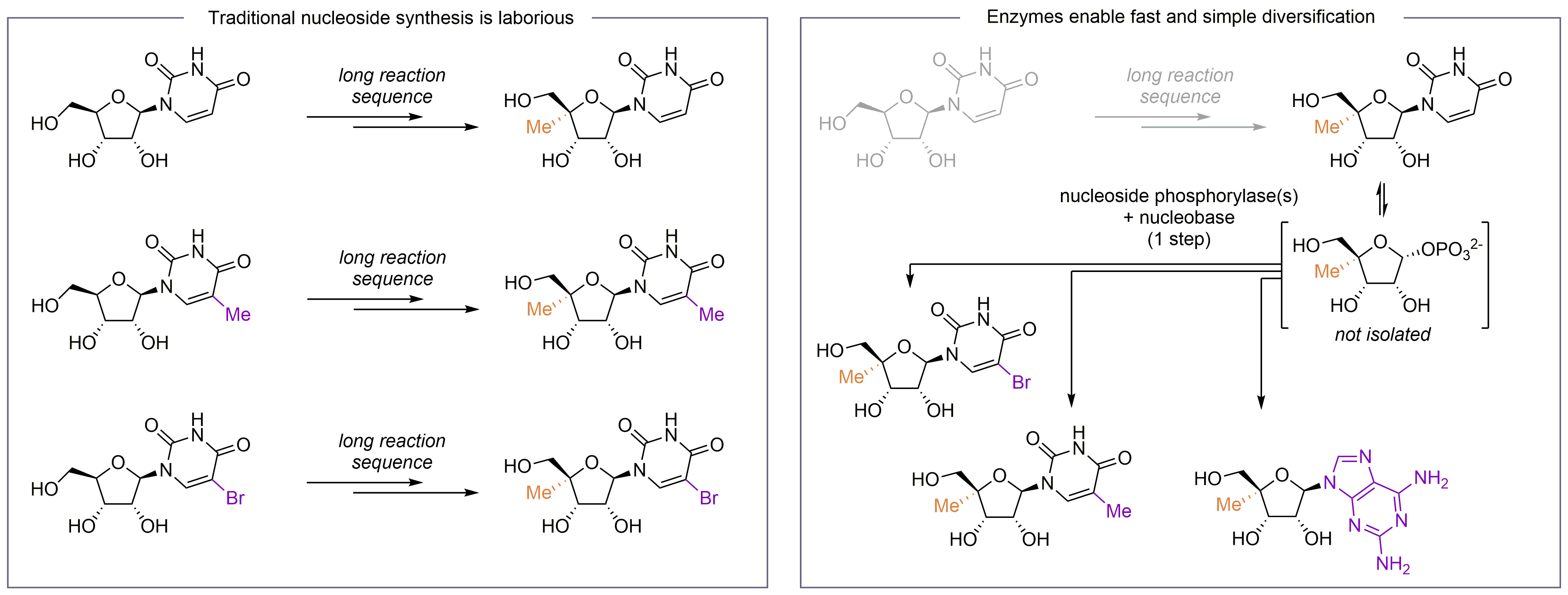
Fig. 1: Nucleoside diversification. Conventional approaches for the introduction of additional substitutions in a nucleoside analogue typically require partial or complete re-synthesis, which is a laborious and inefficient process. Nucleoside phosphorylases readily enable a nucleobase exchange on a modified sugar scaffold. Thus, access to range of nucleoside analogues only requires a single modified precursor.
However, this versatility and the overall great demand for nucleoside analogues stands in striking contrast to their synthetic availability. Nucleoside analogues are complex molecules, densely decorated with functional groups, and often very laborious to make [1]. Chemical syntheses of nucleoside analogues carrying sugar modifications typically involve more than ten steps and, more importantly, exhibit a pronounced lack of divergence. In many cases, the introduction of additional substitutions entails complete or partial re-synthesis of the target compound (Figure 1). This presents a less than ideal situation for biological screening campaigns or drug development purposes. Thus, a diversification strategy that could rapidly attach different nucleobases on a modified sugar scaffold would be a valuable complement to existing techniques.
Researchers from three German universities have explored a biocatalytic solution to this problem, using thermostable nucleoside phosphorylases. Nucleoside phosphorylases are highly conserved enzymes that play a central role in nucleoside catabolism. Natively, they catalyse the reversible phosphorolytic cleavage of nucleosides to obtain the corresponding nucleobase and ribose-1-phosphate. The latter can either be transferred back into the primary carbon metabolism or be engaged in nucleoside synthesis by running the phosphorolysis in the reverse direction – hence attaching a different nucleobase on the sugar scaffold. The combined process of detaching a nucleobase from the sugar (phosphorolysis) and re-attaching a different nucleobase (reverse phosphorolysis or glycosylation) is commonly termed a transglycosylation [2,3]. Conveniently, this reaction sequence effectively turns one nucleoside into another, via a sugar phosphate intermediate that never needs to be isolated (Figure 1).
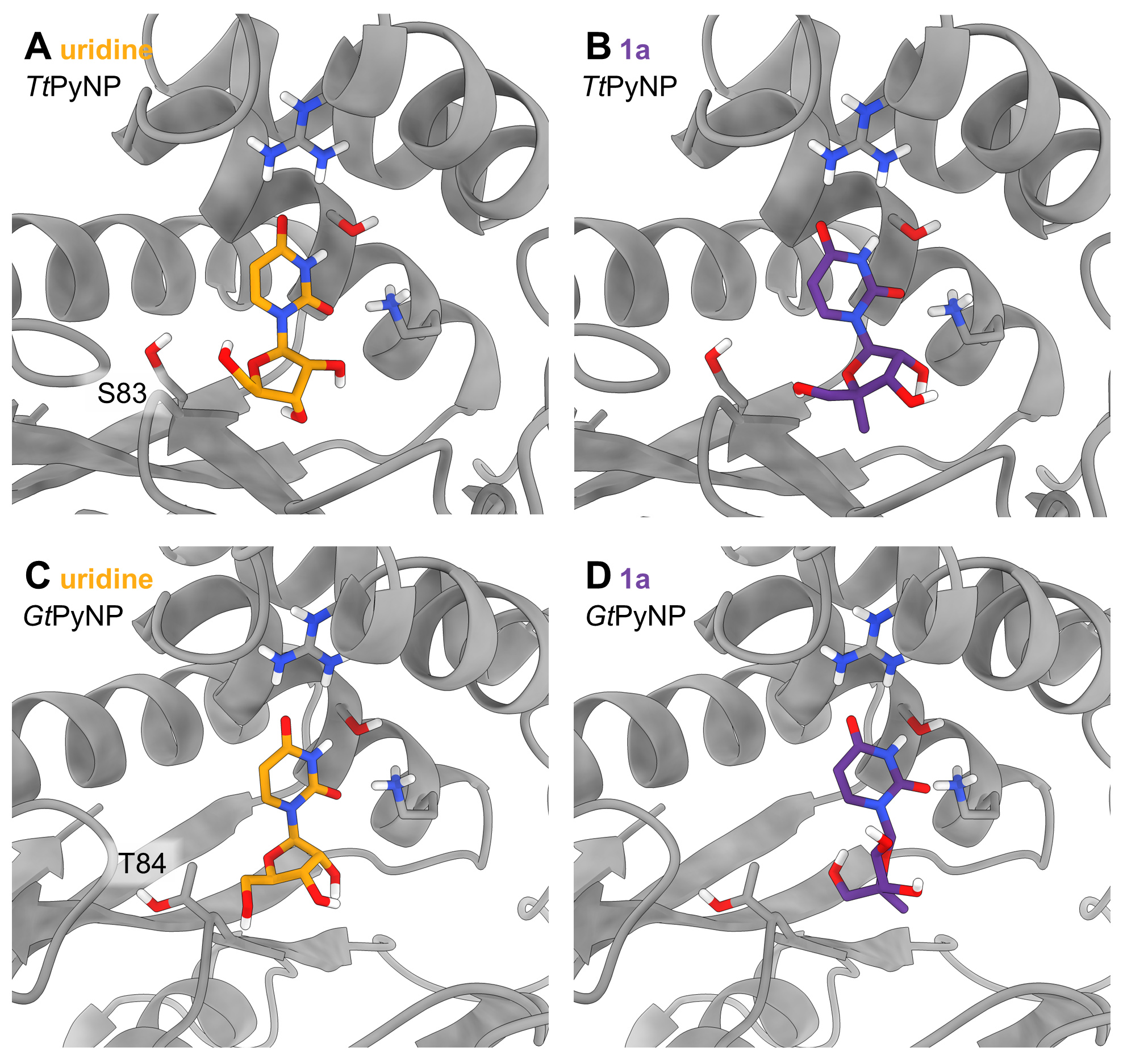
Fig. 2: Docking of the native substrate uridine (orange carbon atoms) and the key methylated analogue 1a (purple carbon atoms) points to subtle steric changes in the back of the active site, which act as a gatekeeper for productive substrate binding. In the inactive enzyme (GtPyNP), the sterically demanding nucleoside analogue preferably adapts an unproductive twisted conformation. The introduction of a threonine-serine substitution in the active site of the previously inactive GtPyNP resulted in a small but measurable activity.
A study led by researchers from the Technische Universität Berlin describes how screening [4] of a panel of promiscuous wild-type pyrimidine nucleoside phosphorylases identified a candidate for phosphorolysis and transglycosylation reactions with nucleoside analogues carrying an alkylated sugar scaffold. The pyrimidine nucleoside phosphorylase from Thermus thermophilus (TtPyNP) is extremely stable [5] and selectively delivers an alkylated sugar phosphate, which can be transformed into a series of other nucleoside analogues. Experimental thermodynamic studies revealed that principles of thermodynamic control [3] could predict the maximum yields obtained in these reactions with striking accuracy, which proved empowering for further reaction optimisation. Remarkably, the wild-type TtPyNP already exhibited synthetically useful activity without the need for further protein engineering, while all other tested nucleoside phosphorylases were inactive with the key substrate. Crystallographic data and docking studies suggest that a subtle threonine-serine substitution at the back of the active site enabled the enzyme to accommodate the sterically more demanding nucleoside analogue (Figure 2). In support of this hypothesis, introduction of this substitution in a previously inactive nucleoside phosphorylase (GtPyNP) installed a small but measurable activity.
Collectively, this research lays the foundation for the systematic exploration and characterisation of nucleoside phosphorolysis equilibrium systems and provides structural data and insights, which will facilitate the engineering of nucleoside phosphorylases for the diversification of nucleoside analogues.
Principal publication and authors
Diversification of 4′-Methylated Nucleosides by Nucleoside Phosphorylases, F. Kaspar (a,b), M. Seeger (c), S. Westarp (a,b), C. Köllmann (d), A.P. Lehmann (d), P. Pausch (e), S. Kemper (f), P. Neubauer (a), G. Bange (e), A. Schallmey (c), D.B. Werz (d), A. Kurreck (a,b), ACS Catal. 11, 10830-10835 (2021); https://doi.org/10.1021/acscatal.1c02589
(a) Chair of Bioprocess Engineering, Technische Universität Berlin (Germany)
(b) BioNukleo GmbH, Berlin (Germany)
(c) Institute for Biochemistry, Biotechnology and Bioinformatics, Technische Universität Braunschweig (Germany)
(d) Institute of Organic Chemistry, Technische Universität Braunschweig (Germany)
(e) Center for Synthetic Microbiology (SYNMIKRO), Philipps-University Marburg (Germany)
(f) Institute for Chemistry, Technische Universität Berlin (Germany)
References
[1] F. Kaspar et al., Green Chem. 23, 37-50 (2021).
[2] F. Kaspar, R.T. Giessmann et al., Adv. Synth. Catal. 362, 867-876 (2020).
[3] F. Kaspar, R.T. Giessmann et al., ChemBioChem 21, 1428-1432 (2020).
[4] F. Kaspar et al., ChemBioChem 21, 2604-2610 (2020).
[5] F. Kaspar et al., ChemBioChem 22, 1385-1390 (2021).



