- Home
- News
- Spotlight on Science
- Jumping crystals:...
Jumping crystals: the structural details of their impressive motility revealed by in situ X-ray powder diffraction
08-12-2014
Certain crystals, when taken through a phase transition, are dramatically self-actuated and jump abruptly to release the internal strain, leaping to distances many times their own size. This rare phenomenon of “jumping crystals”, known as the thermosalient effect, was noticed in the 1980s and has remained poorly understood ever since. The analysis of high-resolution powder diffraction data collected in situ helped to reveal the driving force behind this crystal locomotion.
Share
Solids capable of self-actuation, jumping, exploding, bending or changing their physical appearance upon heating, present a novel platform for clean and effective conversion of thermal energy to mechanical work [1–3]. These dynamic crystals are promising candidates for artificial muscles, smart medical devices and implants, actuators, sensors, electromechanical devices, among others. Performance optimisation of such materials necessitates understanding the underlying physical process at the atomic scale. In a short article from 1983, Etter and Siedle reported that when crystals of (phenylazophenyl)palladium hexafluoroacetylacetonate (PHA) are heated, they “literally fly off the hot stage” [4] (videos of crystals jumping via www.nature.com). This observation was to become the first documented report of the thermosalient (TS) effect. Although this system could help to understand how ordered matter responds to extreme internal pressures, the relationships between the structural, mechanistic and kinematic aspects have remained unknown.
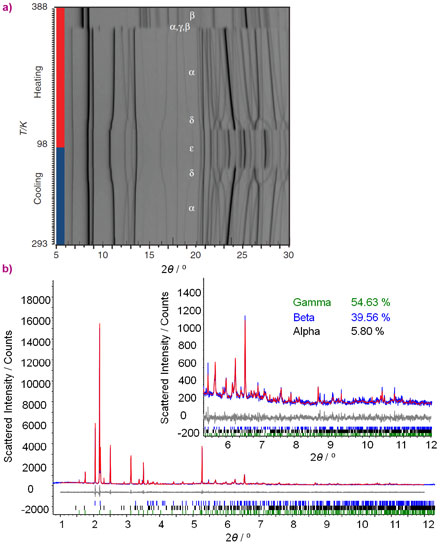
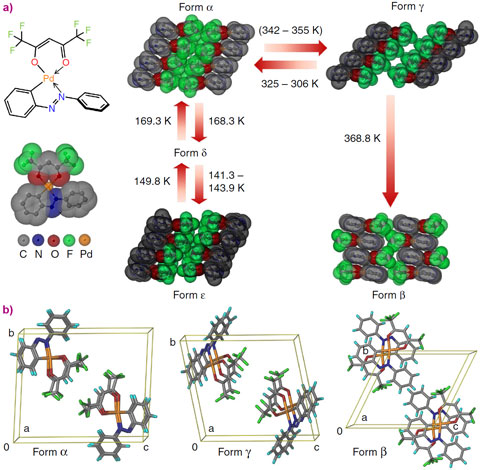
When single crystals of PHA jump, they severely increase their mosaicity and disintegrate, thwarting the solution of the crystal structure by single crystal diffraction. The macroscopic disintegration of single crystals is not an obstacle for X-ray powder diffraction (XRPD); thus, this was the method of choice to investigate the PHA system further. In situ laboratory XRPD studies revealed the presence of as many as 5 polymorphs in the 98 – 388 K temperature window—a record for the largest number of polymorphs of an organometallic compound (Figure 1a). Combined crystallographic, thermogravimetric, microscopic and spectroscopic analyses revealed that, when heated, crystals of form alpha undergo two subsequent phase transformations. The first one, alpha–to–gamma, is the TS-active transition, responsible for the crystal jumping, while the second one, the gamma–to–beta transition, is TS-inactive. Determination of the crystal structure of form gamma was crucial to understand the TS effect in PHA. This phase was found to exist in a three-way mixture with alpha and beta. Moreover, the relative amounts of the phases were found to change under isothermal conditions. Heating to a certain temperature (for example 358 K) resulted in a three-phase mixture. Under constant temperature, the amount of beta phase gradually increased on account of the transformation of alpha and a two-phase (gamma and beta) mixture was eventually obtained. Thereafter, gamma gradually transformed into beta. When dealing with crystal structures of metastable species, it is necessary that the data are collected at the same or preferably lower time-scale relative to the lifespan of the metastable structures. Laboratory XRPD data suitable for crystal structure solution require hours for collection, and thus conventional X-ray sources could not be applied to this system. High-resolution powder diffraction data were collected at beamline ID31 (now ID22), with a time-resolution of 3 minutes per powder pattern, making the TS phase transition amenable to structural investigation (Figure 1b).
Figure 2 shows the phase transitions and crystal structures of four of the five polymorphs of PHA. During the TS phase transition, the layers of molecules in form alpha slide over each other, resulting in slanting of the columns of stacked molecules, which generates immense internal strain, released in the form of forceful jumps of the crystals and transformation into form gamma. In the second, a non-TS transition, alternating layers of molecules in form gamma slide in opposite directions and interdigitate into the thermodynamically stable head-to-tail stacking pattern of form beta. Detailed crystallographic study of this intriguing jumping-crystal phenomenon was only possible by using synchrotron radiation. The PHA research project demonstrates the necessity of acquiring high-resolution diffraction data in future studies of novel materials for energy conversion.
Principal publication and authors
Colossal positive and negative thermal expansion and thermosalient effect in a pentamorphic organometallic martensite, M.K. Panda (a), T. Runčevski (b), S.C. Sahoo (a), A.A. Belik (c), N.K. Nath (d), R.E. Dinnebier (b), P. Naumov (a), Nature Communications 5, 4811 (2014).
References
[1] Ž. Skoko, S. Zamir, P. Naumov, J. Bernstein, J. Am. Chem. Soc. 132, 14191 (2010).
[2] S.C. Sahoo, S.B. Sinha, M.S.R.N. Kiran, U. Ramamurty, A. Dericioglu, M.C. Reddy, P. Naumov, J. Am. Chem. Soc. 135, 13843 (2013).
[3] S.C. Sahoo, M. Panda, N.K. Nath, P. Naumov, J. Am. Chem. Soc. 135, 12241 (2013).
[4] M.C. Etter, A.R. Siedle, J. Am. Chem. Soc. 105, 641 (1983).
Top image: Snapshots (taken at frequency 10 ms) of the kinematic effect of hopping of PHA crystals.