- Home
- News
- Spotlight on Science
- RIXS reveals where...
RIXS reveals where TiO2 nanoparticles store electrons after plasmon-induced electron transfer from Au
03-06-2015
The photo-catalytic performance of TiO2 nanoparticles can be improved by attaching metal particles that have strong absorption bands in the visible region of the light spectrum. An optically excited surface plasmon in Au metal particles may trigger a charge transfer to the TiO2 particle thus improving its ability to catalyse chemical reactions. In this work, the modification of the TiO2 electronic structure following the charge transfer was studied in order to address the question of where the TiO2 particle stores the additional charge.
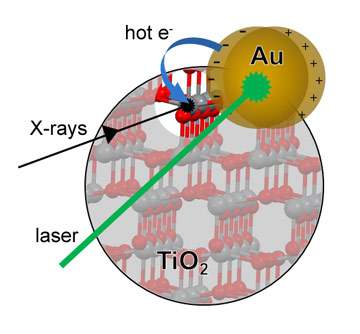
TiO2 is ubiquitous in our everyday life. It is the most widely used white pigment and it finds application as sunscreen and UV blocking filter in cosmetics because of its efficient absorption of UV light. At the beginning of the ‘70s, TiO2 was found to have catalytic properties and was used to split water electrochemically [1]. Since then, the idea of exploiting TiO2 for catalysis fuelled by solar light has encouraged researchers to improve the performance of TiO2 under illumination with visible light. The energy absorption can be enhanced by using a different material to absorb the visible light which then transfers the energy/charge to the catalytically active TiO2 (Figure 1). Metal nanoparticles are very efficient light absorbers because of the localised surface plasmon resonance (LSPR) which is a collective excitation of conductive electrons stimulated by specific wavelengths in the visible range. Devices based on plasmonic nanoparticles and TiO2 show indeed good performance [2] and a better understanding of the charge transfer process can drive innovation in the field.
We studied the charge transfer from Au nanoparticles to N-doped TiO2 at an atomic level by selectively probing the electronic structure of Ti in the TiO2/Au systems under continuous laser illumination. The spectral differences between measurements with and without the laser light demonstrate how the transfer of electrons from Au to TiO2 affects the electronic and atomic structure around Ti atoms. This information can be exploited to deduce where the injected electrons are trapped.
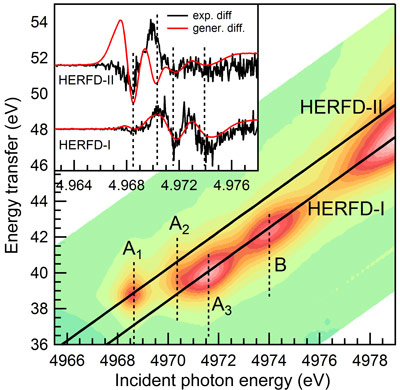
Resonant inelastic X-ray scattering (RIXS) data were collected at the Ti site using beamline ID26. The applied RIXS process involves the excitation of a 1s electron into the lowest unoccupied orbitals (K absorption pre-edge) and a subsequent decay of a 3p electron (Kβ decay). The distribution of the spectral intensity by varying the incident energy and the energy transfer is called the RIXS plane (Figure 2). The line plots representing diagonal cuts through the RIXS plane are referred to as high energy resolution fluorescence detection (HERFD) absorption scans. The pre-edge features in the RIXS plane are nicely separated into two components lying on parallel HERFD lines. Figure 2 shows the RIXS plane and the two cuts intercepting A3 and B (HERFD-I) and A1 with a weak contribution from A2 (HERFD-II). This separation reflects different contributions of Ti atomic orbitals to the spectral features: more delocalised orbitals (Ti p) contribute to features lying on HERFD-I while more localised orbitals (Ti d), to features lying on HERFD-II.
We investigated the effect of electron injection by acquiring HERFD-I and HERFD-II with and without laser illumination. In both cases, small but marked differences in the pre-edge (inset Figure 2) and edge region were detected. The spectral differences induced by the laser on HERFD-I can be modelled by a shift of the spectrum acquired without the laser by -0.8 eV (Figure 2). However, the experimental difference observed in HERFD-II cannot be explained by a simple spectral shift but arises from a modulation of the intensities of A1 and A2 (inset of Figure 2). This twofold behaviour of the pre-edge features upon laser irradiation indicates that the charge remains trapped in Ti p-orbitals and the signal coming from these Ti atoms is red-shifted compared to unaffected Ti atoms. The intensity variations in A1 and A2 are not consistent with a local reduction of Ti4+ to Ti3+ but can be explained with structural modifications in distorted or low-coordinated Ti sites found on the surface of the nanoparticle. We thus propose that the trapping is favoured by p-d orbital mixing caused by local distortion of the TiO6 octahedra at the surface.
Excited states with long lifetimes (>µs) dominate the spectral difference because the X-ray probing and the laser irradiation are continuous [3]. The electrons trapped in the vicinity of Ti atoms thus belong to long-lived states near the surface. Oxidation and reduction of molecules on the surface of TiO2 particles require charge carrier lifetimes in the µs to ms range. The presence of chemically active, long-lived excited states that were found in distorted Ti sites at the surface is thus fundamental to understand the photocatalytic activity.
Principal publication and authors
Probing long-lived plasmonic-generated charges in TiO2/Au by high-resolution X-ray absorption spectroscopy, L. Amidani (a,b), A. Naldoni (c), M. Malvestuto (d), M. Marelli (c), P. Glatzel (b), V. Dal Santo (c), F. Boscherini (a), Angewandte Chemie Int. Ed. 54, 5413 (2015).
(a) Department of Physics and Astronomy, University of Bologna (Italy)
(b) ESRF
(c) CNR-Istituto di Scienza e Tecnologie Molecolari, Milano (Italy)
(d) Elettra-Sincrotrone Trieste (Italy)
References
[1] A. Fujishima, K. Honda, Nature 238, 37 (1972).
[2] S. Mubeen, J. Lee, N. Singh, S. Kraemer, G.D. Stucky, M. Moskovits, Nat. Nanotechnol. 8, 247 (2013).
[3] M.H. Rittmann-Frank, C.J. Milne, J. Rittmann, M. Reinhard, T.J. Penfold, M. Chergui, Angew. Chem. Int. Ed. 53, 5858 (2014).
Top image: Electrons originating from the de-excitation of surface plasmons on Au nanoparticles are injected into TiO2 and remain trapped on surface Ti distorted sites. The effect of the extra-charge is detected in RIXS that is able to separated Ti d- and p-orbitals.