- Home
- News
- Spotlight on Science
- Microbeam diffraction...
Microbeam diffraction gives new insights into the working of lithium-ion battery electrodes
23-09-2015
Phase transitions in Li-ion electrode materials during charge and discharge are decisive for battery performance, limiting high-rate capabilities, and play a crucial role in the cycle life of Li-ion batteries. However, the difficulty to probe the phase nucleation and growth within individual grains is hindering fundamental understanding and progress. Synchrotron microbeam diffraction has uncovered the rate dependent phase transition mechanism within individual particles of LiFePO4, a key Li-ion electrode material. At low charge and discharge rates, well-defined nanometre thin-plate shaped domains coexist and transform much more slowly than was commonly assumed. As the charge and discharge rate increases the phase boundaries become diffuse and this speeds up the transformation rates of individual grains. Direct observation of the transformation of individual grains reveals that local current densities significantly differ from previous assumptions, giving new insights into the working of Li-ion battery electrodes and their potential improvements.
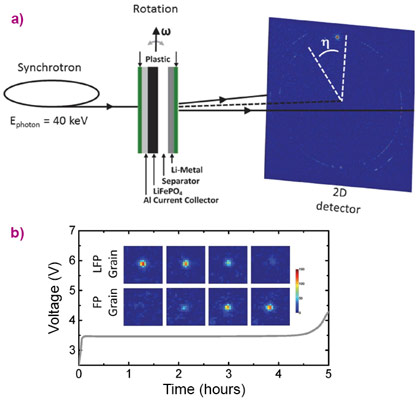
The overall phase transformation behaviour of Li-ion battery electrode materials is well known. However, even for well-studied electrode materials such as LiFePO4, it is unknown how the transformation proceeds within individual grains and how it is distributed over the many grains present in an electrode. This is crucial information for future battery improvement because the local conditions, such as the local current density, govern the Li-ion battery cycle life and rate performance. Using the microbeam at ID11, operando X‑ray diffraction was employed to monitor the transformation of many individual LiFePO4 particles inside an Li-ion battery electrode from slow to fast battery charging and discharging rates, as shown schematically in Figure 1a. The diffraction results revealed the cycling-rate dependent phase transition mechanism, shown in Figure 2 and Figure 3, within individual electrode grains. Measuring the transformation process of individual LiFePO4 particles allowed the first quantification of local current densities, i.e. the current per surface area of individual grains.
Due to both the sub-micron size and brightness of the X-ray beam at ID11, the diffraction rings that are conventionally observed from powder diffraction break up into individual spots, each originating from individual nanosized LiFePO4 grains. By using a specially designed working battery this allowed the evolution of the (200) reflection to be monitored for individual LiFePO4 particles during charge and discharge, as shown in Figure 1. The gradual disappearance of the (200) LiFePO4 and appearance of the (200) FePO4 diffraction peaks reflect the first-order phase transition when Li-ions are removed from the LiFePO4 electrode.
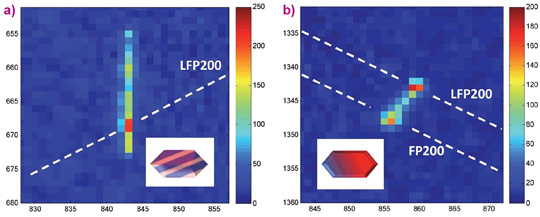
A surprising observation is that a significant fraction of the reflections during slow charge and discharge appear as streaks as illustrated by the reflections in Figure 2a, rather than symmetric peaks as shown in Figure 1b. The considerable one-dimensional broadening implies platelet-shaped domains of both FePO4 and LiFePO4 as shown in Figure 2a, where the plate thickness is inversely proportional to the length of the streaks. The position of the streaks on the (200) diffraction ring (characterised by the azimuth angle h indicated in Figure 1a) in combination with the streak orientation allows us to determine the crystallographic orientation of the platelet-shaped domains in the LiFePO4 grains. This showed that in these LiFePO4 particles there is a distribution of FePO4/LiFePO4 interfaces ranging from coherent to complete loss of coherence in the (001) direction.
When the charge and discharge rate is increased, the streaked reflections narrow and disappear, translating into a decrease in number of platelet domains and an average increase in platelet domain thickness upon increasing the current. For example, Figure 2b shows the appearance of a double peaked reflection at a fast charging rate. The appreciable intensity between the LiFePO4 (200) and FePO4 (200) end-member indicates a continuous distribution of intermediate compositions, reflecting a diffuse interface over the full length scale of the 140 nm grain. This proves that the recently observed bypassing of the first-order phase transition [1] occurs within a single grain, rationalising the excellent battery performance of this electrode material.
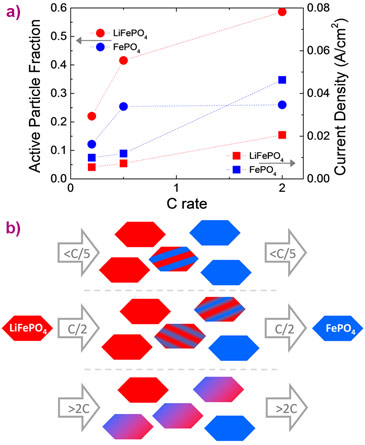
Measuring the transformation process of individual LiFePO4 particles allowed the first quantification of local current densities in Li-ion battery electrodes under realistic charge and discharge conditions, as shown in Figure 3a. The local current densities are larger than expected if all electrode active material is assumed to participate but smaller than that expected from a particle-by-particle dominated mechanism. The charge and discharge rate dependence of the local current density, which was derived from the average transformation times of individual grains, is a crucial parameter for electrode design that can help to prevent the occurrence of hotspots and fractures in electrodes. The increase in the local current density is a direct consequence of the faster transformation of individual particles upon increasing the charge and discharge rate. In combination with the increasing fraction of actively transforming particles, the faster transformation mechanism is responsible for supplying the larger currents when increasing the charge and discharge rates.
Principal publication and authors
Direct view on the phase evolution in individual LiFePO4 nanoparticles during Li-ion battery cycling, X. Zhang (a), M. van Hulzen (a), D.P. Singh (a), A. Brownrigg (a), J.P. Wright (b), N.H. van Dijk (a) and M. Wagemaker (a), Nature Communications 6, 8333 (2015).
(a) Department of Radiation Science and Technology, Delft University of Technology (The Netherlands)
(b) ESRF
References
[1] X. Zhang, M. van Hulzen, D.P. Singh, A. Brownrigg, J.P. Wright, N.H. van Dijk and M. Wagemaker, Nano Letters 14, 2279-2285 (2014).
Top image: Tracking diffraction spots from individual grains in a battery while charging or discharging reveals the phase transformations that occur.