- Home
- News
- Spotlight on Science
- Combining operando...
Combining operando X-ray techniques to study lithium ion batteries
13-09-2016
Operando X-ray tomographic microscopy (SRXTM) and scanning X-ray diffraction (SXRD) have been combined for the first time to study dynamic processes in a lithium ion battery. This combination of techniques allowed the lithiation processes to be tracked throughout the electrodes on different length scales: the dynamic distribution of crystallographic phases was analysed using SXRD, while SRXTM data was used to probe volumetric changes and understand the interplay of different kinetic limitations.
Synchrotron X-ray tomographic microscopy (SRXTM) has become an increasingly popular imaging technique to non-invasively study lithium ion battery electrodes upon electrochemical operation. Depending on the resolution, this technique allows retrieval of 3D information about the electrode microstructure and even individual particle shapes.
Lithiation kinetics and electrode degradation are linked to crystallographic changes in the active materials. In particular, for alloying materials, such as silicon, the formation of different crystallographic phases upon lithiation is accompanied by large volumetric changes of the active material, which affect transport kinetics in the electrode microstructure, as well as the mechanical integrity of the electrode. However, these structural changes on the scale of the crystal lattices of the active materials are not accessible using SRXTM alone.
Here, we show that correlations between dynamic effects at the atomic and the microstructure length scales may be studied by combining SRXTM and SXRD within a single operando experiment. This approach has the additional benefit that many battery materials may be analysed including those that are either (1) weakly X-ray attenuating and therefore pose a challenge to attenuation-based tomography or are (2) amorphous and therefore pose a challenge to diffraction techniques.
The battery sample studied in this report was assembled from a silicon powder electrode, a glass fibre separator immersed in electrolyte, and a metallic lithium counter electrode. During the operando experiment, the cell was cycled so that lithium ions are inserted in the silicon electrode. Silicon was chosen as the active material because, with a theoretical specific charge capacity close to ten times that of graphite, it is of high interest for the design of next generation lithium-ion battery electrodes. Furthermore, the electrochemical reactions cause observable effects on different length scales, which is ideally suited for the combined analysis approach: distortions of the crystal lattices may be tracked using SXRD, while volumetric changes of the electrode may be tracked using SRXTM.
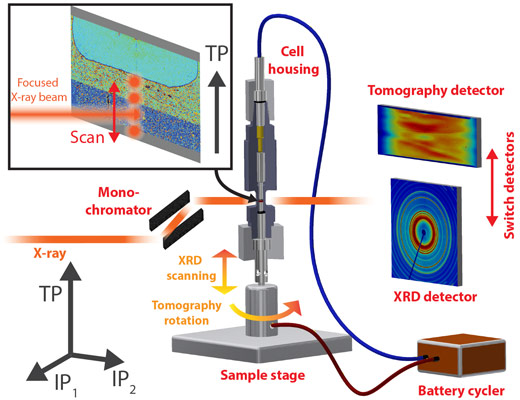
Figure 1 shows the experimental setup at beamline ID11. For a tomographic scan, monochromatic X-rays penetrate the rotating sample and the projections that are acquired every fraction of a degree are reconstructed to a 3D image dataset. For SXRD, the X-ray beam is focused to a spot and a series of diffraction patterns is collected at different depth positions (through-plane direction (TP)) in the silicon electrode (see Figure 1 inset). These two measurements are repeated continuously and in alternating order as the silicon electrode is lithiated.
 |
|
Figure 1. Sketch of the experimental setup. |
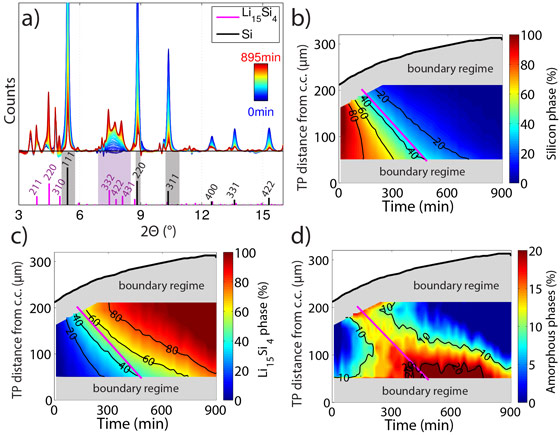
The resulting diffraction patterns from the SXRD measurements at a fixed position in the silicon electrode are shown as a function of time in Figure 2a. During the course of lithiation, the silicon peaks (grey shaded) vanish due to electrochemical amorphisation, while a new phase identified as fully lithiated silicon (Li15Si4, magenta shaded) appears. Based on the recorded diffraction patterns, the scientists quantified the relative phase fractions of pristine silicon (Figure 2b), Li15Si4 (Figure 2c) and intermediate amorphous phases (Figure 2d) within the silicon electrode as a function of time and the TP direction. Lithiation of the silicon electrode starts at the interface with the separator and then gradually progresses through the electrode towards the interface with the current collector.
 |
|
Figure 2. (a) X-ray diffraction patterns and deduced maps of the phase fractions of (b) pristine silicon, (c) Li15Si4 and (d) amorphous phases. |
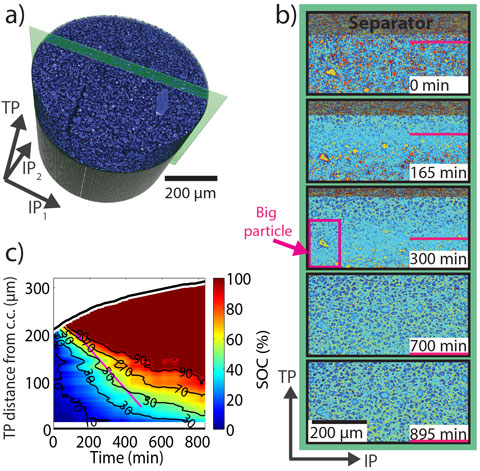
The tomographic measurements reveal the volumetric expansion of the electrode and also enable extraction of the state of charge distribution throughout the silicon electrode. Virtual cuts through the silicon electrode at different times (Figure 3b) along the green plane depicted in Figure 3a show the silicon particles become lighter because their mass density and accordingly their X-ray attenuation coefficient decreases when they become lithiated. The image contrast is thus a measure of the local state of charge. We exploited this relation to quantitatively map out the state of charge distribution in the electrode as a function of through-plane direction and time (Figure 3c). The results are in good agreement with the findings from SXRD (Figure 2b-c).
 |
|
Figure 3. (a) Rendering of and (b) cuts through the silicon electrode. (c) Quantification of the state of charge distribution in the silicon electrode. |
In this study, we were able to demonstrate the power of combined X-ray techniques and also highlight an interesting phenomenon that occurs in high capacity electrodes. Even though the electrode studied was quite porous, which would typically imply that the kinetics of charge transport are not limited by ionic diffusion, the silicon particles, with their high capacity, act as a sink for lithium ions. Before lithium can diffuse deep into the electrode, the particles at the top lithiate almost entirely, leading to a pronounced charge gradient in the electrode. This highlights that ultra-high capacity electrode materials might require different electrode designs if incorporated into today’s lithium ion batteries.
Principal publication and authors
Combining operando synchrotron X-ray tomographic microscopy and scanning X-ray diffraction to study lithium ion batteries, P. Pietsch (a), M. Hess (a), W. Ludwig (b), J. Eller (a) & V. Wood (a). Scientific Reports 6, 27994 (2016); doi: 10.1038/srep27994.
(a) Laboratory of Nanoelectronics, ETH Zurich (Switzerland)
(b) ESRF



