- Home
- News
- Spotlight on Science
- Ancestral-sequence...
Ancestral-sequence reconstruction unveils the structural basis of function in mammalian flavin-containing monooxygenases
24-01-2020
Flavin-containing monooxygenases are xenobiotic degrading enzymes ubiquitous in all organisms. Using ancestral sequence reconstruction, three ancient mammalian flavin-containing monooxygenases were characterised, unveiling the structural basis for their function.
Xenobiotic detoxification is an ancient process pursued by all organisms [1]. Over the course of evolution, organisms have equipped themselves with a range of intricate proteins, employed to disable and combat a myriad of toxins and xenobiotics. One class of these enzymes is known as flavin-containing monooxygenases (FMOs). These enzymes reside in the membranes of the endoplasmic reticulum and are involved in phase I of xenobiotic degradation, oxidising a plethora of substrates, including many pharmaceuticals [2]. Despite their broad substrate profiles and drug metabolising capacity depicting them as formidable catalysts, structural information regarding these enzymes and our understanding of substrate uptake and conversion was lacking.
Humans possess five FMOs that are dispersed throughout many tissues including the liver and lungs. An intriguing feature among the FMOs is that FMO5 pursues Baeyer-Villiger monooxygenase activity, as opposed to FMOs 1-3 which all involve soft nucleophiles, typically possessing sulfur and/or nitrogen atoms, abstracting an electrophilic oxygen from a key flavin-(hydro)peroxy intermediate [2,3]. The aim of the research described here was to employ ancestral sequence reconstruction to unravel the underlining evolutionary events that resulted in the ‘explosion’ of FMO genes in tetrapods (four-limbed organisms), whilst confirming whether ancestrally-derived FMOs possessed similar biocatalytic attributes observed in the extant constructs. Three ancient mammalian FMOs were synthesised, denoted as AncFMO2, AncFMO3-6 and AncFMO5, representing the ancestral proteins of the FMO isoforms: 2, 3 and 6 collectively, and 5, respectively.
The reconstructed phylogeny portrayed jawed vertebrate FMOs as monophyletic and derived from a single common ancestor. Furthermore, the earliest mammalian ancestor had already encoded the five FMO paralogues resulting from four major gene-duplication events. The AncFMOs were successfully transformed and expressed in E. coli and proved enzymatically competent. Steady-state kinetics experiments involving an NADPH-depletion spectrophotometric assay were used to confirm that the enzymes exhibit biocatalytic activity and substrate profiles akin to their extant successors. These findings indicate that over millennia, FMOs had conserved typical catalytic features of class B flavoprotein monooxygenases.
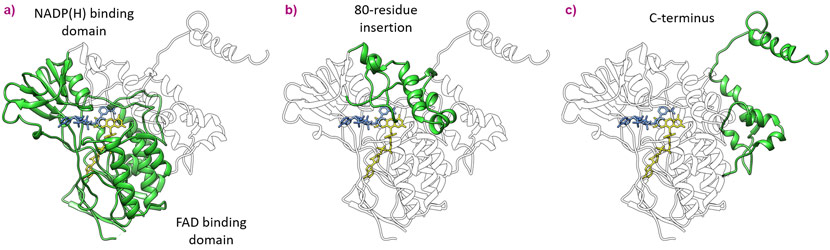
The ancestral FMOs were then purified and subjected to detergent and crystallisation screenings. Remarkably, all three proteins were successfully crystallised and first datasets were collected for AncFMO2 at beamlines ID30B and the automated beamline ID30A-1 (MASSIF-1). Fortunately, using an insect FMO baring a sequence identity of 32%, resolutions between 2.7 and 3.2 Å were sufficient to solve the structure by molecular replacement. Furthermore, structure determination was greatly facilitated by density averaging thanks to the asymmetric unit of the crystals of AncFMO2 possessing four protein molecules. These ancient enzymes conveyed both well-conserved and novel structural features (Figure 1).
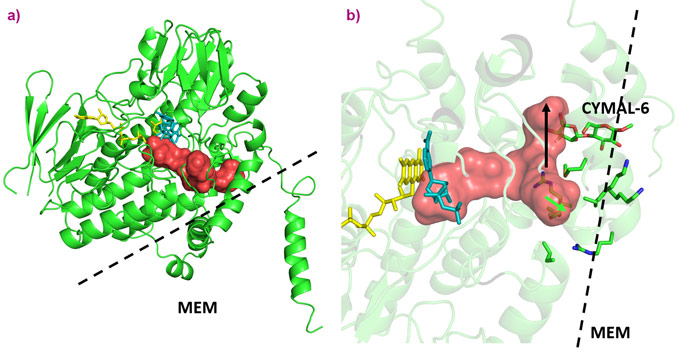
The active sites among the ancestral FMOs portrayed a well-conserved architecture with very little change in cavity size or amino acid decor. The Baeyer-Villiger monooxygenase activity, unique to FMO5, is likely derived from a key switch between a glutamate residue, observed in AncFMO2 and AncFMO3-6, with a histidine residue observed in the vicinity of the FAD ring. Unlike the glutamate residue, the histidine can employ a hydrogen-bond donating role to stabilise the concomitant negatively-charged Crigee intermediate [4]. A substantial difference however, between the mammalian FMO structure and their soluble counterparts seen in bacteria, fungi and insects is that the active site is in a closed cavity shielded from the solvent via an 80-residue insertion (Figure 1). Additionally, this insertion feeds the active site by orchestrating a series of tunnels that bind and penetrate the membrane whilst also providing additional exit/entrance points into the solvent (Figure 2). The key features that govern the substrate preferences among the AncFMOs could not be delineated but the high similarity among the active sites alludes towards the tunnels representing the key properties dictating and controlling substrate influx.
Thermal stability assays demonstrated that the AncFMOs possessed heightened stabilities with respect to human FMOs, reaching melting temperatures up to 22°C higher. Intriguingly, they were more stable in the presence of oxidised coenzyme, NADP+, a feature unobserved for the human FMOs. Moreover, sequence similarities between AncFMOs and their respective human FMO were very high, between 82 and 92%, with the differing residues shown on the surface of enzyme and not directly influencing cofactor binding or substrate uptake. Together, these changes should confer significantly increased stability on the protein. The thermostable, yet functionally and structurally conserved proteins, delivered by ancestral sequence reconstruction are reliable models for understanding the function and drug metabolising ability of the human FMOs whilst also highlighting the technique as a tool for protein crystallisation.
Principal publication and authors
Ancestral-sequence reconstruction unveils the structural basis of function in mammalian FMOs. C.R. Nicoll (a), G. Bailleul (b), F. Fiorentini (a), M.L. Mascotti (c), M.W. Fraaije (b) and A. Mattevi (a), Nat. Struct. Mol. Biol. 27, 14–24 (2020); doi: 10.1038/s41594-019-0347-2.
(a) Department of Biology and Biotechnology “Lazzaro Spallanzani”, University of Pavia (Italy)
(b) Molecular Enzymology, Groningen Biomolecular Sciences and Biotechnology Institute, University of Groningen (The Netherlands)
(c) IMIBIO-SL CONICET, Facultad de Química Bioquímica y Farmacia, Universidad Nacional de San Luis (Argentina)
References
[1] W.B. Jakoby & D.M. Ziegler, The enzymes of detoxication, J. Biol. Chem. 265, 20715–20718 (1990).
[2] S.K. Krueger & D.E. Williams, Mammalian flavin-containing monooxygenases: structure/function, genetic polymorphisms and role in drug metabolism, Pharmacol. Ther. 106, 357–387 (2005).
[3] F. Fiorentini et al., Baeyer-villiger monooxygenase FMO5 as entry point in drug metabolism. ACS Chem. Biol. 12, 2379–2387 (2017).
[4] E. Romero, J.R.G. Castellanos, G. Gadda, M.W. Fraaije, & A. Mattevi, The same substrate, many reactions: oxygen activation in flavoenzymes, Chem. Rev. 118, 1742–1769 (2017).