- Home
- News
- Spotlight on Science
- Pathways of hepatitis...
Pathways of hepatitis B virus capsid assembly
26-08-2020
The assembly pathways of hepatitis B virus capsid were resolved using time-resolved SAXS, simulations, thermodynamic analysis, and maximum information entropy optimisation. The earliest steps of the reaction, controlled by the association free energy between capsid proteins, dictate the assembly pathway.
Viruses are the most abundant biological entities on our planet. Their structure, composed of only few components, presents an ideal example of a biologically functional, self-assembled structure. Viruses encapsulate and protect their genetic materials in a protein shell called capsid. Minimising the amount of viral genome coding structural proteins, capsids are comprised of many copies of a small number of proteins, often only one, usually in a spherical or helical arrangement. This minimalistic design, held together by weak protein-protein interactions, supports an extremely efficient assembly process, resulting in a monodisperse, stable structure.
Half of the known virus families have icosahedral capsids (polyhedrons containing 20 triangular faces, with five-fold symmetry around each vertex). While the structures of many capsids are known, their assembly and disassembly processes are still poorly understood.
Capsids can have a hundred or even thousands of subunits, consequently there are a huge number of possible intermediates, and many more potential assembly pathways. Yet, assembly is rapid and with high fidelity. Experimental kinetic data at high spatial and temporal resolutions covering the early stages of assembly, are critical for resolving the underlying mechanism of virus assembly/disassembly.
Recently, the assembly pathways of the hepatitis B virus (HBV) capsid were observed. HBV causes about 270 million cases of chronic infection, leading to about 880,000 deaths each year from liver disease and cancer. HBV has an icosahedral capsid composed of homodimeric core protein (Cp). In vivo assembly involves spontaneous nucleation to form empty particles, comprising 90% of the particles present during infection. Recombinant capsid protein assembly domain, Cp149 (the first 149 residues of Cp), assembles in vitro into empty capsids, identical to the capsids isolated from virus-expressing cells.
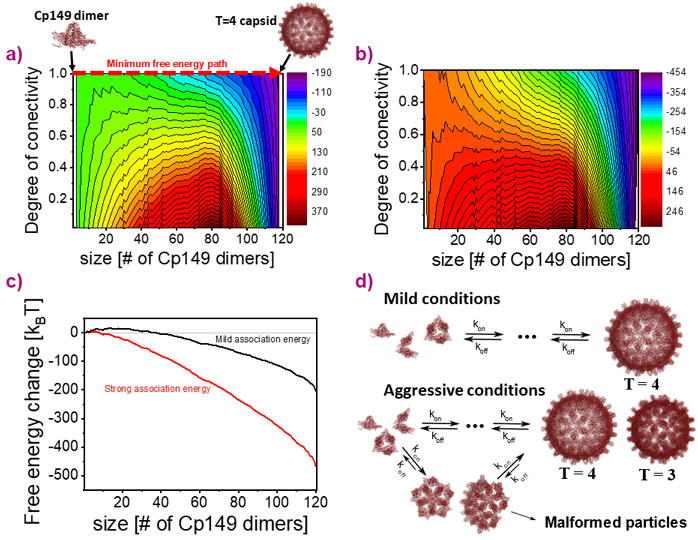
HBV capsids are composed of 90 (capsid symmetry T = 3) or 120 (T = 4) Cp149 dimers. There are an estimated 1030 intermediates on the assembly path from dimer to complete capsid. Hence, resolving the HBV capsid assembly mechanism is an ill-posed problem. To identify assembly pathways, graph theory was used to represent the structure of HBV intermediates. Umbrella sampling of Monte Carlo simulations was then performed to create a comprehensive library of unique assembly intermediates [1]. Using the software D+ (scholars.huji.ac.il/uriraviv/software/d-software) [2], the atomic model of each member in the library was generated and its solution small angle X-ray scattering (SAXS) curve was computed. Intermediates were selected from this library based on a grand canonical free energy landscape of HBV capsid that had been calibrated with experimental SAXS data HBV capsid assembly reactions at equilibrium [1,3].
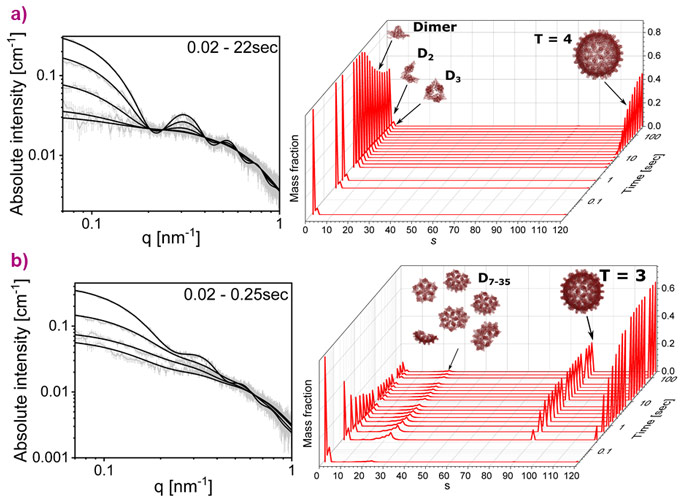
Using the time-resolved SAXS setup of ID02, the real-time assembly of HBV capsids was tracked (Figure 1). To rigorously analyse the data with minimal bias, maximum informational entropy optimisation analysis was applied with prior distributions, derived from the calibrated grand canonical free energy landscape at the onset of the assembly reactions (Figure 2) [3]. Equilibrium-based prior knowledge filtered out the most stable intermediates that are likely to make a significant contribution to the assembly reaction.
The pathway of HBV capsid assembly was discovered to be dictated by early intermediates. At mild, biologically-relevant, dimer-dimer association free energy, assembly started after a lag phase of 10 s and appeared to be a two-state reaction with Cp149 dimers and complete T = 4 capsids. Other intermediates did not accumulate to detectable amounts. The grand canonical free energy landscape predicts a relatively high and broad barrier to initiating assembly that prevents the accumulation of intermediates, following which the energy decreased towards the full capsid with no local minima. This landscape is consistent with multiple reversible steps, allowing the reaction to follow the minimum free-energy path, at which the most stable and compact intermediate structures are dominant. Under aggressive assembly conditions, where the dimer-dimer association free energy was about 1 kBT stronger, the reaction was much faster, and the dimer concentration decreased without any measurable lag phase. A diverse array of intermediates, containing between 7 and 35 dimers accumulated within the first 250 milliseconds, after which capsids with two different radii were detected (corresponding to capsid symmetries of T=4 and T=3). After the first second, the dimer concentration was still sufficient to support assembly of T=4 capsids by either slow elongation of the intermediates or establishing new 'capsid assembly lines' [3].
Our findings show that small changes in the dimer-dimer association free energy control the earliest steps of the reaction, which dictate the subsequent assembly pathway, and may provide strategies for understanding, regulating, and designing assembly of protein cages.
Principal publication and authors
Intermediate Structures Dictate the Pathway of Capsid Assembly, J. Am. Chem. Soc. 142, 7868–7882 (2020); doi: 10.1021/jacs.0c01092.
(a) Institute of Chemistry and the Center for Nanoscience and Nanotechnology, The Hebrew University of Jerusalem (Israel)
(b) Molecular and Cellular Biochemistry Department, Indiana University, Bloomington (United States)
References
[1] Asor R, Selzer L, Schlicksup CJ, Zhao Z, Zlotnick A, Raviv U., ACS Nano 13, 7, 7610–7626 (2019).
[2] Ginsburg A, Ben-Nun T, Asor R, Shemesh A, Fink L, Tekoah R, Levartovsky Y, Khaykelson D, Dharan R, Fellig A and Raviv U., J. Appl. Cryst. 52, 219-242 (2019).
[3] Asor R, Schlicksup CJ, Zhao Z, Zlotnick A, Raviv U., J. Am. Chem. Soc. 142, 17, 7868–7882 (2020).