- Home
- News
- Spotlight on Science
- X-ray imaging with...
X-ray imaging with sandpaper enables advanced 3D virtual histology
18-09-2020
Three-dimensional images of a kidney with unprecedented quality have been obtained with the simple, cost-effective addition of sandpaper as an optical element to an X-ray imaging beamline. The results demonstrate the potential of X-ray speckle-based tomography for quantitative 3D virtual histology.
Biological soft tissue and other low-density materials can be visualised quantitatively and in 3D with a higher image quality than ever before by using a recently developed X-ray imaging technique that makes use of a piece of sandpaper as an optical element.
The need for 3D visualisation and characterisation of biological soft tissue has driven an increasing interest in X-ray-based virtual histology. Conventional histology, the current gold standard for tissue visualisation, struggles to deliver 3D data with isotropic resolution, as it is based on physical slicing of the sample and scanning of the slices with a light microscope. Expanding histology to the third dimension is a critical step for a more accurate analysis of the sizes, shapes and interrelationship of sample features. This will contribute to an improved assessment of tissue pathologies and a better understanding of organ function.
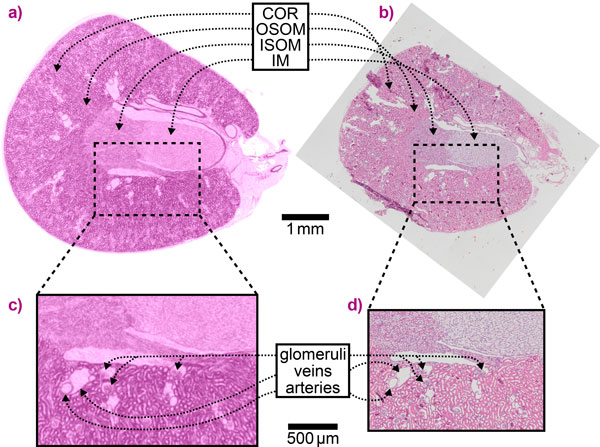
An X-ray imaging technique known as speckle-based imaging (SBI) has been further developed to enable quantitative three-dimensional virtual histology (3DVH) of unstained, hydrated biological tissue at unprecedented image quality. Using beamline ID19, X-ray images of a mouse kidney were obtained with extremely high contrast that have been validated with conventional histology results, as shown in Figure 1.
SBI [1] is based on the X-ray phase-contrast signal, which exploits the refraction of X-rays in the sample as a contrast mechanism, leading to a significantly higher sensitivity to small density variations than conventional absorption X-ray imaging. SBI was first demonstrated at the ESRF [2] and simultaneously at Monash University [3] and has since seen great interest from the X-ray imaging community thanks to its extremely robust, simple and cost-effective implementation. The only required addition to a conventional X-ray imaging setup is a piece of sandpaper. Scattering and interference of X-rays from the sandpaper grains lead to the formation of a speckled intensity pattern, known as near-field speckles, in the detector plane. X-ray refraction in a sample results in a local displacement of these speckles, which can be computationally retrieved and converted to a phase-contrast signal. SBI combined with tomography, i.e. taking projections of the sample at different viewing angles, allows for the reconstruction of the 3D density distribution within the sample. Among the different data acquisition and analysis methods developed for SBI, the unified modulated pattern analysis (UMPA) used for the data presented here has shown particularly high performance and flexibility [4].
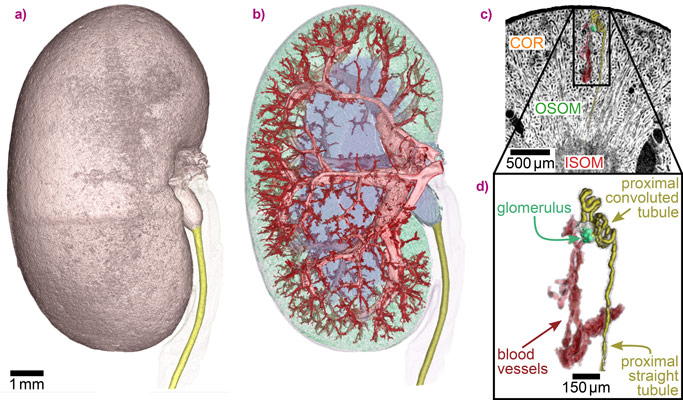
The high sensitivity of SBI makes it possible to visualise even minute density differences in soft tissue at high contrast without the use of staining agents. This enables the straightforward and accurate segmentation of features in a sample, such as the complex blood vessel network of the kidney in this study (Figure 2). Furthermore, one single renal nephron, the functional unit of the kidney, could be extracted from the SBI data, which is a critical step towards gaining a better understanding of organ function and dysfunction. The 3D information on the sample is complemented with the quantitative mass density values that SBI delivers in addition to the structural information.
With its high sensitivity, quantitative character and simple setup, X-ray speckle-based imaging has great potential as an imaging method in fundamental biomedical research and clinical histopathology to extend and complement conventional histology. Moreover, the method is also promising for applications in other fields that rely on visualising small density differences such as materials science and palaeontology.
The results of this research are illustrated by the following movie: Summary of the 3D virtual histology of the mouse kidney.
Principal publication and authors
X-ray phase tomography with near-field speckles for three-dimensional virtual histology, Zdora M.-C. (a,b,c), Thibault P. (a,b), Kuo W. (d,e), Fernandez V. (f), Deyhle H. (b), Vila-Comamala J. (g), Olbinado M.P. (h,i), Rack A. (i), Lackie P.M. (j), Katsamenis O.L. (k), Lawson M.J. (j), Kurtcuoglu V. (d,e,l), Rau C. (b), Pfeiffer F. (m,n), Zanette I. (a), Optica 7, 1221-1227 (2020); doi: 10.1364/OPTICA.399421.
(a) School of Physics & Astronomy, University of Southampton (United Kingdom)
(b) Diamond Light Source, Didcot (United Kingdom)
(c) Department of Physics & Astronomy, University College London (United Kingdom)
(d) The Interface Group, Institute of Physiology, University of Zurich (Switzerland)
(e) National Centre of Competence in Research, Kidney.CH, Zurich (Switzerland)
(f) Imaging and Analysis Centre, Natural History Museum, London (United Kingdom)
(g) Institute for Biomedical Engineering, ETH Zurich (Switzerland)
(h) Paul Scherrer Institute, Villigen PSI (Switzerland)
(i) ESRF
(j) Clinical and Experimental Sciences, Faculty of Medicine, University of Southampton (United Kingdom)
(k) μ-VIS X-Ray Imaging Centre, Faculty of Engineering and Physical Sciences, University of Southampton (United Kingdom)
(l) Zurich Center for Integrative Human Physiology, University of Zurich (Switzerland)
(m) Chair of Biomedical Physics, Department of Physics and Munich School of BioEngineering, Technical University of Munich, Garching (Germany)
(n) Department of Diagnostic and Interventional Radiology, Klinikum rechts der Isar, Technical University of Munich (Germany)
References
[1] Zdora M.-C., J. Imaging 4, 60 (2018).
[2] Berujon S. et al., Phys. Rev. Lett. 108, 158102 (2012).
[3] Morgan K.S. et al., Appl. Phys. Lett. 100, 124102 (2012).
[4] Zdora M.-C. et al., Phys. Rev. Lett. 118, 203903 (2017).
Top image: Simultaneous visualisation of tissue structure and blood vessels of a mouse kidney obtained with X-ray speckle-based phase tomography.