- Home
- News
- Spotlight on Science
- Mapping the minutiae:...
Mapping the minutiae: Insights into the growth of graphene with liquid metal catalyst
23-01-2023
Growing graphene on a molten metal surface may be the best way to get a high-quality, defect-free product in commodity quantities. X-ray techniques at beamline ID10 were combined with machine learning to characterise the interface between the 2D material and a liquid copper catalyst with unprecedented sub-angstrom precision.
Share
Graphene is one of the strongest materials on earth, even more so than diamond. It has a wide range of applications in transportation, electronics and medication, owing to its unique mechanical, electric and optical properties, which can be attributed to its equally unique structure. The creation of a one atom-layer thickness 2D graphene film was recognised with a Nobel Prize in 2010. Despite its widespread popularity and applications, its large-scale commercialisation has been hindered due to the defects formed while synthesising it.
Recently, liquid metal catalysts such as copper (Cu) have been used to synthesise extremely high-quality graphene, almost without any defects. The liquid surface of the catalyst is perfectly flat, which is thought to be the reason behind graphene's highly crystalline structure. The fundamental problem with understanding liquid metal catalysts in detail is the microscopic processes occurring at the surface, which are not detectable by conventional experiments and computational approaches. This is because of the harsh conditions needed for graphene's growth and the dynamic liquid interface.
Set against this backdrop, an EU-funded collaboration of researchers used molecular dynamics simulations based on machine learning (ML) algorithms and synchrotron radiation experiments to study this system with unprecedented accuracy. This team was led by theoreticians from the Fritz Haber Institute in collaboration with experimentalists from the ESRF as well as the Universities of Aarhus in Denmark and Leiden in the Netherlands.
The interface between graphene and liquid Cu is very delicate and key to understanding how a high-quality product can be grown. The team were eager to investigate whether ML-assisted simulations would predict the interface in accordance with the synchrotron experiments. Both in-situ and in-silico techniques were used to characterise the adsorption height of the graphene monolayer above liquid Cu.
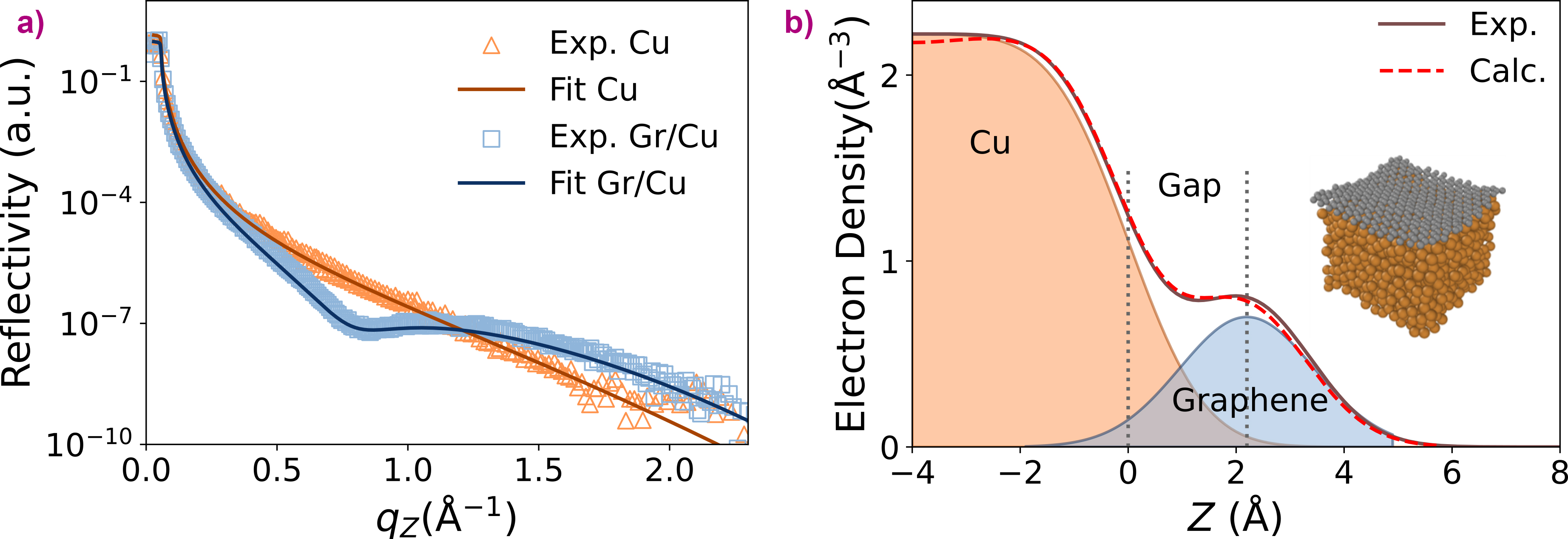
Fig. 1: a) Experimental X-ray reflectivity curves taken in situ from the bare liquid Cu (orange symbols) and the liquid Cu covered with a graphene (Gr) layer grown by chemical vapour deposition (blue symbols). The solid curves show the fit used to extract the electron density along the vertical direction. b) Electron density profiles from experiment (brown) and simulation (red) as well as the corresponding species-resolved profiles of Cu (orange) and graphene (grey). Vertical dotted lines denote the Cu density inflection point and the peak of the graphene profile. The ‘gap’ is derived as the distance between these two heights. The simulation cell is shown by the inset.
The experimental results were obtained using in-situ synchrotron X-ray reflectivity at beamline ID10 (Figure 1a), which needed to work with the curvature of the liquid surface, imposing an additional degree of experimental complexity. To build the computational model, the team trained the ML potentials, specifically, moment tensor potentials, using density functional theory reference data. This helped to obtain large-scale molecular dynamics simulations while leveraging the efficiency of ML algorithms.
The team then determined the adsorption height of monolayer graphene above liquid Cu, known as ‘the gap’, experimentally and computationally (Figure 1b). It was found that the experimental value of the gap was 2.2 Å, whereas the computational or theoretical value was 2.119 Å. With a less than 0.1 Å difference, the results demonstrated an almost quantitative agreement between theory and experiment.
The results show that ML potentials trained with first principles are a powerful approach that can determine the gap with sub-angstrom potential, while being in good agreement with the experimental value. This is a prerequisite for future detailed mechanistic investigations. Surprisingly, the computational techniques reveal that the interaction of graphene with solid and liquid Cu is chemically identical, thus adding to the mystery of the superior synthesis from the liquid metal state. These powerful insights can serve for future understanding of a seamless graphene synthesis to develop next-generation electronics.
Principal publication and authors
Graphene at Liquid Copper Catalysts: Atomic-Scale Agreement of Experimental and First-Principles Adsorption Height, H. Gao (a), V. Belova (b), F. La Porta (b), J. Santiago Cingolani (c), M. Andersen (d), M. Saedi (e), O.V. Konovalov (b), M. Jankowski (b), H.H. Heenen (a), I.M.N. Groot (e), G. Renaud (f), K. Reuter (a), Adv. Sci. 9, 2204684 (2022); https://doi.org/10.1002/advs.202204684
(a) Fritz-Haber-Institut der Max-Planck-Gesellschaft (Germany)
(b) ESRF
(c) Technische Universität München (Germany)
(d) Aarhus University (Denmark)
(e) Leiden University (The Netherlands)
(f) Université Grenoble Alpes (France)
| About the beamline: ID10 |
|
ID10 is a soft interfaces and coherent scattering beamline composed of two end-stations. End-station EH1 is for high-resolution X-ray scattering and surface diffraction on liquid and solid interfaces, combining multiple techniques in a single instrument. This allows investigation of the structure and self-organisation processes at surfaces, interfaces and in thin films both in-plane and normal to the film. End-station EH2 is for coherent small-angle X-ray scattering, X-ray photon correlation spectroscopy, which allows the study of slow in-equilibrium and out-of-equilibrium dynamics in disordered or modulated materials on timescales beyond the reach of the inelastic (X-ray or neutron) techniques, and coherent diffraction, allowing for the high-resolution imaging of biomineral hierarchical structures, porous semiconductor materials, mineral nanocrystals and nanostructures, biological cells, etc. |