- Home
- News
- Spotlight on Science
- X-ray nanodiffraction...
X-ray nanodiffraction reveals impact of lattice imperfections on battery cathode materials
05-02-2024
High-voltage spinels hold promise as cathode materials for next-generation Li-ion batteries, offering a combination of high energy density and rapid (de)lithiation kinetics. Operando X-ray nanodiffraction provides insights into the structural transformations that occur within spinel single crystals during electrochemical delithiation.
Beyond its use as a gemstone, the spinel crystal structure is found in a number of metal oxides. One notable example is the high-voltage spinel LiMn1.5Ni0.5O4 (LMNO). This oxide exhibits unique properties, making it an attractive candidate for cathode materials in Li-ion batteries. It acts as a host structure for lithium ions, enabling their reversible insertion and extraction during battery charging and discharging. High operating voltage and rapid (de)lithiation kinetics are among the most appealing properties of these Co-free oxides. Moreover, unlike most other transition metal-based cathode materials, they can be readily grown into micrometre-sized single crystals. Single-crystal cathode materials are expected to show higher mechanical strength than cathodes consisting of nanosized particles. This enhanced strength may prevent cracking, subsequent pulverisation, and capacity loss, as supported by studies [1,2]. Due to these improved properties, single crystal cathodes are anticipated to significantly enhance the cycling life of batteries, enabling thousands of charge–discharge cycles without significant capacity loss.
The dynamic processes of lithiation and phase transition dynamics in battery cathode materials remain incompletely understood due to the complexities involved in distinguishing between interparticle and intra-particle heterogeneity. Larger crystallites, such as in the case of LMNO, further complicate this matter by producing intricate lithium concentration gradients within the active material. Effectively controlling these microstructural features is crucial for maximising the durability and charge rate capabilities of single-crystal cathode technologies. However, imaging the movement of lithium ion intercalation at the nanoscale is an extremely challenging task.
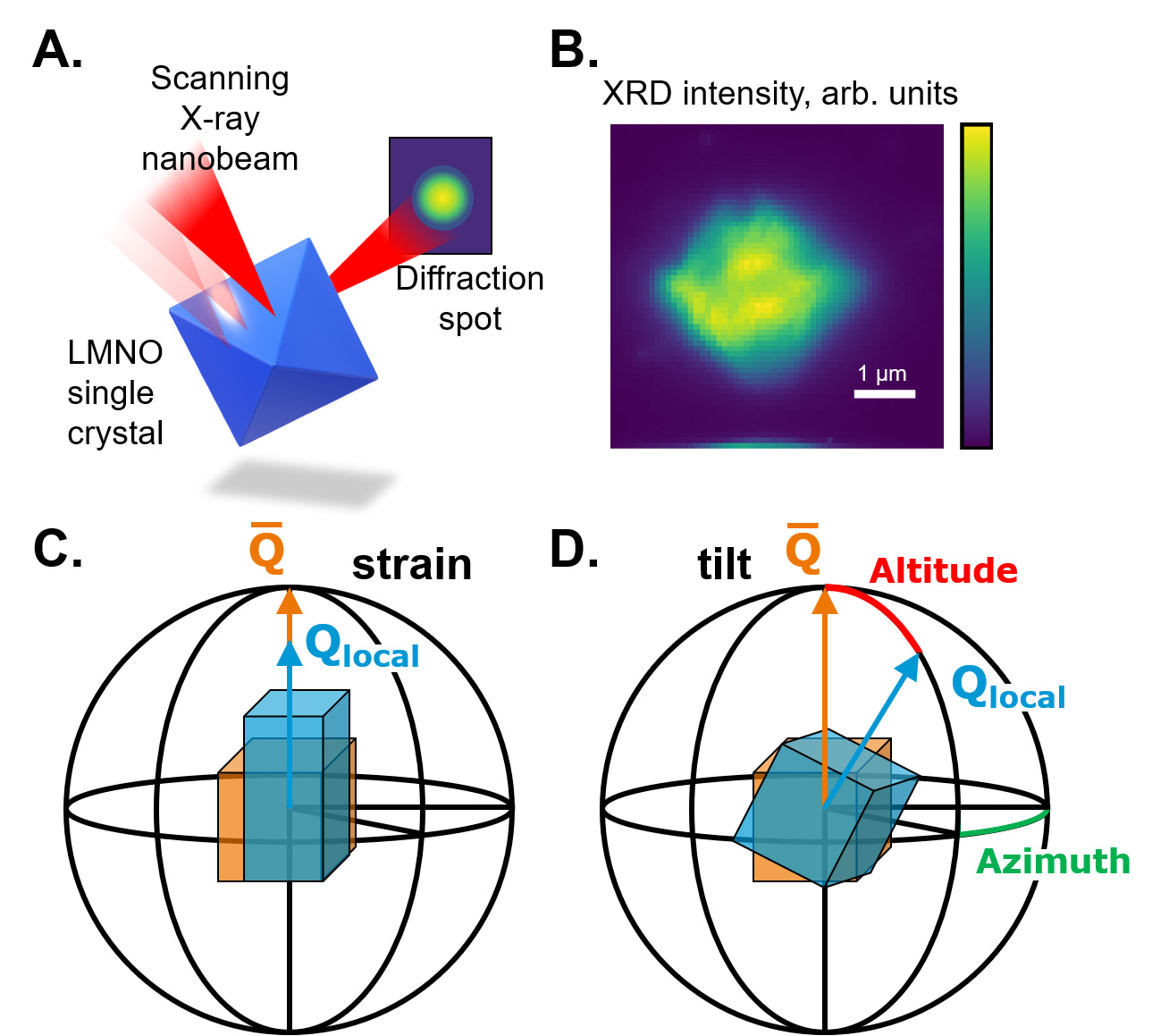
ESRF beamline ID01 is specialised in the nano-imaging of crystalline materials. This work utilised scanning X-ray diffraction microscopy (SXDM) at ID01, an imaging technique that utilises a nano-focused X-ray beam to scan across a sample (Figure 1a), measuring diffraction intensity at each position on the crystal (Figure 1b). The resulting maps are highly sensitive to local lattice strain (Figure 1c) and crystallographic rotation/tilt of the measured planes (Figure 1d).
Fig. 1: a) Overview of the scanning X-ray diffraction microscopy (SXDM) technique. b) A measured map of the diffracted intensity across an LMNO crystal. SXDM is sensitive to changes of the magnitude of the scattering vector Q (c), corresponding to changes of the d-spacing and to its relative angular deviation (d), related to lattice misorientation.
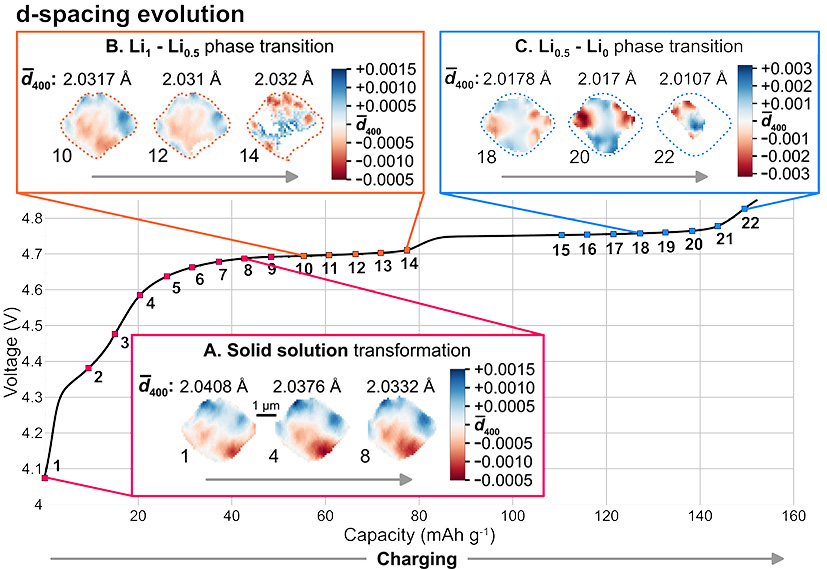
The lattice evolution within the cathode particle is directly linked to the lithium extraction due to significant changes in the lattice unit cell. Prior operando diffraction studies have revealed that the delithiation of LMNO during charging involves multiple steps associated with different phase transition dynamics [3]. However, SXDM provides an unprecedent view into the sub-100-nm intraparticle lattice heterogeneities, enabling the investigation of these mechanisms at the nanoscale.
Fig. 2: Evolution of d-spacing maps of an LMNO single crystal during charging. Three different stages of lattice transformations are highlighted.
The initial step involves a solid–solution transformation, characterised by a gradual decrease in the lattice constant due to Li-ion extraction. SXDM enabled the observation of metastable domains forming within the particle (Figure 2a, highlighted in pink). These domains do not appear to be associated with the reaction front between the lithiated and delithiated phases, as predicted by current phase transition theory. Instead, unusually persistent strain gradients within the single crystals suggest that the shape and size of solid–solution domains are templated by lattice defects, which play a crucial role in guiding the entire delithiation process.
Subsequent to the solid–solution transformation, two consecutive two-phase transitions occur, characterised by abrupt lattice parameter reductions and the formation of well-defined domains within the particle (Figure 2b and c, highlighted in orange and blue). These transitions are attributed to distinct changes in the oxidation state of nickel atoms: Ni2+ → Ni3+ for the first transition, and Ni3+ → Ni4+ for the second. Morphology analysis, strain distribution patterns and tilt boundary observations revealed that these phase transitions involve different mechanisms. The first transition exhibits a more heterogeneous mechanism, with highly localised delithiation. In contrast, the second transition proceeds through a homogeneous “core-shell” transformation. In both cases, lattice defects tend to act as nucleation points, facilitating the phase transformation dynamics on the surface of the crystal.
These findings highlight the significance of lattice imperfections, which are often present in cathode materials used in batteries. Controlling these defects will be critical for understanding the influence of doping, cation-ordering and coatings, and will offer a solution for mitigating structural degradation in high-voltage spinel active materials, propelling progress towards commercially viable durability.
Principal publication and authors
Defects and nanostrain gradients control phase transition mechanisms in single crystal high-voltage lithium spinel, I. Martens, (a), N. Vostrov (a), M. Mirolo (a), S.J. Leake (a), E. Zatterin (a), X. Zhu (b), L. Wang (c), J. Drnec (a), M.-I. Richard (a,d), T.U. Schülli (a), Nat. Commun. 14, 6975 (2023); https://doi.org/10.1038/s41467-023-42285-4
(a) ESRF
(b) College of Materials Science and Engineering, Changsha University of Science and Technology, Changsha (China)
(c) Nanomaterials Centre, School of Chemical Engineering, and Australian Institute of Bioengineering and Nanotechnology, University of Queensland, Brisbane (Australia)
(d) Univ. Grenoble Alpes, CEA Grenoble, IRIG, MEM, NRX, Grenoble (France)
References
[1] J. Langdon & M. Arumugam, Energy Storage Mater. 37, 143-160 (2021).
[2] G. Qian et al., Energy Storage Mater. 27, 140-149 (2020).
[3] H. Komatsu et al., Adv. Energy Mater. 5(17), 1500638 (2015).
| About the beamline: ID01 |
| ID01 is a versatile X-ray diffraction and scattering beamline that delivers beams down to 35 nm. It is dedicated to the investigation of a broad spectrum of crystalline materials, from nanostructures to bulk, with the ability to perform imaging of strain and structure using full-field diffraction imaging, coherent X-ray diffraction methods and/or nanodiffraction. Typical samples range from microelectronic devices to novel metal-organic solar cells or battery electrodes. |