- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Structure of materials
- Discovery of a novel zeolite-like framework in an oxonitridophosphate
Discovery of a novel zeolite-like framework in an oxonitridophosphate
Zeolites are microporous compounds built up from three-dimensionally interconnected TX4 tetrahedra (typically T = Si, Al, P; X = O) with ions or small molecules within their cavities and channels. Some of these inorganic open-framework structures are used as absorbents, e.g. in detergents, and are essential in industrial processes, e.g. ZSM-5 as a catalyst for hydrocarbon isomerisation reactions. Prompted by such achievements and the potential for future application as sensors or optical systems, aluminosilicates and aluminophosphates have been intensively developed since the 1980s with the goal of generating new materials with different pore shapes and sizes. By October 2011, 197 zeolite framework types had been identified [1]. As the variety is already high, it becomes increasingly difficult to find new topologies in the field of classical zeolite chemistry.
To discover novel frameworks, we shifted from purely oxidic systems to nitridic ones where nitrogen atoms bridge the tetrahedra. After the first nitridic zeolite LixH12-x-y+z [P12OyN24-y]Xz (X = Cl, Br) [2] (three letter code NPO), the higher structural flexibility of nitrogen recently lead once more to an unprecedented zeolite-like framework in the oxonitridophosphate Ba19P36O6+xN66-xCl8+x (x ≈ 4.54) whose structure was elucidated using the high-resolution powder-diffraction beamline ID31.
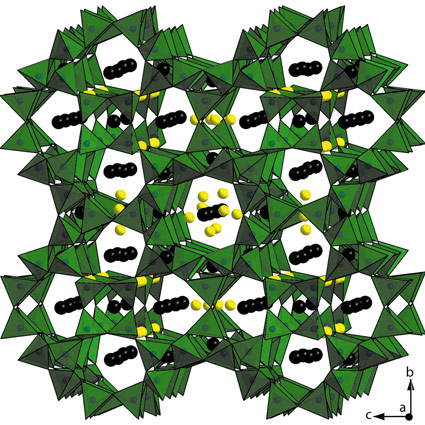
Ba19P36O6+xN66-xCl8+x was obtained by heating phosphoryl triamide OP(NH2)3, thiophosphoryl triamide SP(NH2)3, BaS, and NH4Cl in an evacuated and sealed silica glass ampoule at 750ºC. The identification was challenging as the complex synthesis yielded only microcrystalline products with minor unknown side phases. Hence, we made use of a complementary approach including high-resolution powder X-ray diffraction (HR-PXRD), transmission electron microscopy, and solid-state nuclear magnetic resonance (NMR). After evaluation of several HR-PXRD data sets from different samples measured at ID31 (with λ = 0.4 Å), a primitive cubic unit cell with a = 1342.95 pm or its twofold superstructure (cubic face centred) with doubling of a came into consideration. However, through independent symmetry information gained by electron diffraction and scanning transmission electron microscopy images, the correct lattice parameter as well as the correct space group symmetry could be unequivocally derived. The crystal structure of Ba19P36O6+xN66-xCl8+x (Figure 27) was solved with the charge-flipping algorithm and refined with the Rietveld method in space group Fm3c on the basis of the HR-PXRD data. The structural model was corroborated by 31P double-quantum(DQ) /single-quantum (SQ) and triple-quantum (TQ) /single-quantum (SQ) 2D correlation MAS NMR spectroscopy. 31P{1H} C-REDOR NMR measurements ruled out that stoichiometric amounts of hydrogen are present.
 |
|
Fig. 27: Crystal structure of Ba19P36O6+xN66-xCl8+x (Ba2+ black, Cl- yellow, P(O,N)4 tetrahedra green). Reprinted with permission from J. Am. Chem. Soc. 133, 12069–12078 (2011). Copyright 2011 American Chemical Society. |
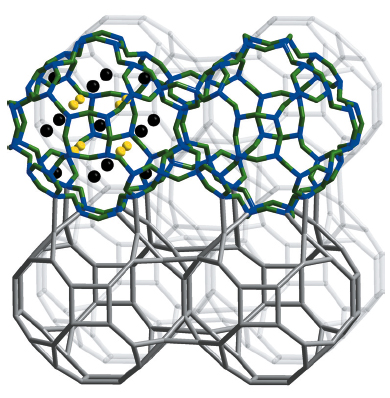
Ba19P36O6+xN66-xCl8+x comprises a framework of all-side vertex sharing P(O,N)4 tetrahedra representing a topology that has been previously predicted as a hypothetical zeolite structure based on SiO2 [3]. The tetrahedra build up slightly distorted cages with 3, 4, and 8 rings (Figure 28). Interconnected through their four rings, these 3846812 cages (superscript numbers mean frequency) generate a network with a low framework density of 14.9 T/1000 Å31 (typical range for zeolites: 12.5–20.2 T/1000 Å3) and a 3D 8-ring channel system with a free diameter of 292 pm. The extra-framework ions Ba2+ and Cl- are incorporated in the cages as well as in the channels. Activation of these ions for ion exchange is the subject of continuing investigations. We now focus on the development of Ba19P36O6+xN66-xCl8+x as a material for applications as it has a high thermal stability (up to 1100°C). To conclude, this novel zeolite-like framework, as the second nitridic zeotype after NPO which is mainly built up from 3-rings, is an important step towards establishing the nitride approach in zeolite chemistry. Since the 15th of October, the network of Ba19P36O6+xN66-xCl8+x has been registered in the database of the International Zeolite Association as NPT, nitridophosphate two [1].
 |
|
Fig. 28: Interconnected 3846812 cages in Ba19P36O6+xN66-xCl8+x; top: drawing with P–N bonding and cage content (left); bottom: representation with only P–P linking. Reprinted with permission from J. Am. Chem. Soc. 133, 12069–12078 (2011). Copyright 2011 American Chemical Society. |
Principal publication and authors
S.J. Sedlmaier, M. Döblinger, O. Oeckler, J. Weber, J. Schmedt auf der Günne and W. Schnick, J. Am. Chem. Soc. 133, 12069–12078 (2011).
Department of Chemistry, Ludwig-Maximilian University Munich (Germany)
References
[1] C. Baerlocher, L.B. McCusker, Database of Zeolite Structures: http://www.iza-structure.org/databases/, accessed January 2012.
[2] S. Correll, N. Stock, O. Oeckler and W. Schnick, Angew. Chem., Int. Ed. 42, 3549–3552 (2003).
[3] M.D. Foster and M.M.J. Treacy, A Database of Hypothetical Zeolite Structures: http://www.hypotheticalzeolites.net, accessed June 2010.



