- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2011
- Structure of materials
- Observing catalysts in multiple dimensions
Observing catalysts in multiple dimensions
Metals and/or metal oxides anchored to porous support materials are the cornerstone of the industrial heterogeneous catalysis industry. These catalysts owe their activity to the stabilisation of the metals/metal oxides in active electronic and/or coordination states, and are often employed in 3D form (known as catalyst bodies) in fixed bed reactors so as to minimise pressure drops across the reactor vessel. The efficiency of the final catalytic system depends on the nature and distribution of these active species within the catalyst body, although this can be controlled at the preparation stage. However, the evolution of the active species from an initial precursor delivered via liquid phase impregnation is often a highly complex process. In recent times analytical techniques have been developed to obtain real time 2D and 3D spatial information on the distribution of chemical species in catalysts during their preparation in order to unravel the physicochemical processes taking place. Such information enables better control of the distribution of the active phase and therefore the overall performance of the catalyst [1]. Recently we employed the technique of time-resolved X-ray diffraction computed tomography (XRD-CT, Figure 46) at beamlines ID15A and ID15B and demonstrated its capability to image a catalyst body as a function of time to follow the preparation process and to study the catalyst as a reaction (CO methanation) takes place [2].
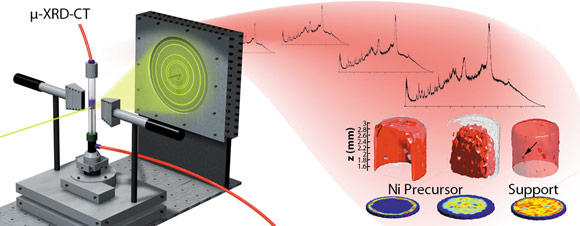 |
|
Fig. 46: In XRD-CT, a series of transmission projection measurements is made at several angles while the sample is exposed to X-ray radiation. A 2D image slice is then reconstructed from these measurements via a suitable algorithm based on the differences in the diffraction signals for the crystalline phases present. |
In the first study, examining the preparation during calcination (in N2) of a Ni supported γ-Al2O3 catalyst body, we observed the formation of the final cubic (fcc) metallic Ni phase via a complex multi-step process involving a number of crystalline intermediate phases. Importantly, two different crystalline phases initially form located at the periphery and in the centre of the body accordingly. Although both phases yielded fcc Ni, the precursor at the periphery did so in one step, while the phase in the centre did so via a 3-4 step process (Figure 47). This appeared to affect the particle size of the fcc Ni phase with the fcc Ni particles in the centre of the pellet larger than those seen at the periphery. This has important implications for the activity/selectivity of these catalyst bodies in a catalytic reaction.
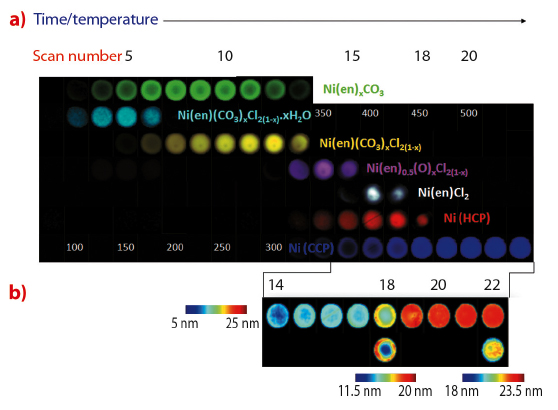 |
|
Fig. 47: Reconstructed 2D images of crystalline components’ features observed in the diffraction patterns as a function of time/temperature during thermal activation of a γ-Al2O3-supported Ni catalyst precursor. a) The colour maps indicate the following distribution of solid-state phases accordingly: precursor 1 (green), precursor 2 (cyan), subsequent breakdown phases associated with precursor 2 (yellow, magenta, white and red), and fcc Ni (blue). b) The thermal colour maps show the variation in crystallite size (nm). When plotted with a common colour axis (top) these indicate the growth of fcc Ni crystallite size as the growth of the phase proceeds. For slices, 18 and 22, the data have been re-plotted with bespoke colour axes (shown beneath). |
In a second recent study, we combined XRD-CT with absorption-CT to study the activity of Ni/γ-Al2O3, to study again the phase evolution process during the processes of calcination and activation, but more importantly, to study such catalyst bodies for the first time under true catalytic conditions. As with the first study, two different, inhomogenously distributed precursors result after the impregnation process, although the breakdown of these precursors resulted in the formation more uniformly-sized Ni nanoparticles. Particles located towards the periphery undergo uncontrolled sintering on exposure to O2 although this appears to be dependent on both the concentration/distribution of the Ni and its form (where it is expected that Ni is present in the sample in both crystalline and diffraction silent forms). Reduction by H2 does not induce further sintering but results in an active fcc Ni-containing catalyst for CO methanation. Operando catalytic measurements performed during CO methanation, demonstrated the crystalline metallic fcc Ni component to be remarkably stable during the duration of reaction (~3 hours), undergoing no change in phase constitution, spatial distribution or average particle size, leading us to conclude that metallic fcc Ni is the active component of the catalyst for CO methanation.
Principal publication and authors
S.D.M. Jacques (a, b), M. di Michiel (c), A.M. Beale (a), T. Sochi (b), M.G. O’Brien (a), L. Espinosa-Alonso (a), B.M. Weckhuysen (a) and P. Barnes (b), Angew. Chem. Int. Ed. 50, 10148, (2011).M.G. O’Brien (a), S.D.M. Jacques (a,b), M. di Michiel (c), P. Barnes (b), B.M. Weckhuysen (a) and A.M. Beale (a) Chem. Sci. 3, 509-523 (2012).
(a) Inorganic Chemistry and Catalysis, Debye Institute for NanoMaterials Science, University of Utrecht (The Netherlands)
(b) UCL Chemistry London/Dept. Crystallography, Birkbeck College, London (UK)
(c) ESRF
References
[1] L. Espinosa-Alonso, A.M. Beale and B.M. Weckhuysen, Acc. Chem. Res. 43, 1279 (2010).
[2] P. Bleuet, E. Welcomme, E. Dooryhee, J. Susini, J.L. Hodeau and P. Walter, Nat. Mater. 7, 468 (2008).



