- Home
- Users & Science
- Find a beamline
- Structure of materials
- ID22 - High resolution powder diffraction beamline
- Science at ID22
- Examples: Dynamic and in-situ studies
Examples: Dynamic and in-situ studies
Dynamic and in-situ studies
Here you will find selected dynamic and in-situ studies carried out on the beamline. Experiments were carried out on ID22 (since 2014), and ID31 (2002 - 2013). Examples include:
Hydrogen cycling between lithium nitride Li3N, lithium imide Li2NH, and lithium amide LiNH2
A potential lightweight hydrogen-storage system involves hydrogen cycling between lithium nitride Li3N, lithium imide Li2NH, and lithium amide LiNH2 via two reversible steps, formally written as
- Li3N + H2 <=> Li2NH + LiH
- Li2NH + H2 <=> LiNH2 + LiH.
The reverse of step (2), the release of 6.5 weight percent of hydrogen from the amide to the imide, is viewed as a two-stage process
2 LiNH2 -> Li2NH + NH3
LiH + NH3 -> LiNH2 + H2.
Three samples that had undergone various cycles of hydrogen adsorption and desorption ex situ were examined on ID31 [1]. As well as a complex mixture of phases identified in sample SI, including the unexpected new phase Li1.15NH1.85 (composition from the refined Li stoichiometry), some remarkable peak shapes were apparent in samples SII and SIII, e.g. figure 1, implying complex microstructural behaviour. The broad Bragg peaks of the dominant cubic Li-N-H phase are highly structured and follow a strain-broadening dependence on diffraction angle, indicative of a pronounced variation in stoichiometry. Detailed modelling of the pattern using TOPAS allowed the range of non-stoichiometry to be evaluated. The variation in stoichiometry of the cubic phase was modelled by 11 uniformly varying phases Li1+xnNH2-xn, where xn = xlow + 0.1n(xhigh - xlow); n = 0 – 10. A linear variation in lattice constant was also assumed; an = ahigh + 0.1n(alow - ahigh). The refined parameters were the lower and upper bounds of the lattice parameter (alow and ahigh) and the stoichiometry, (xlow and xhigh), and the scale factor associated with each phase. Refined stoichiometries range from Li1.08NH1.92 to Li1.65NH1.35 with a weighted average of Li1.52NH1.48 for SII and from Li1.18NH1.82 to Li1.70NH1.30 with a weighted average of Li1.58NH1.42 for SIII.
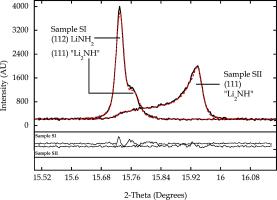
Figure 1. Fit to the 11 peak of the Li amide-imide system using TOPAS.
Apart from illustrating what a truly powerful program TOPAS is in the right hands, and just how much information you can extract from high quality high resolution powder diffraction data, after further consideration of the results the experimenters were able to propose a mechanism for hydrogen storage and release.
This study was part of an extensive program to find new hydride materials [e.g. 2], exploiting the high-throughput capabilities of ID31. The users will come with as many as 200 samples to be measured over a couple of days. In-situ adsorption and desorption experiments are also performed.
[1] A Mechanism for Non-stoichiometry in the Lithium Amide/Lithium Imide Hydrogen Storage Reaction. W. I. F. David, M. O. Jones, D. H. Gregory, C. M. Jewell, S. R. Johnson, A. Walton, and P. P. Edwards, J. Am. Chem. Soc., 129, 1594 (2007).
[2] Synthesis and crystal structure of Li4BH4(NH2)3. P.A. Chater, W. I. F. David, S. R. Johnson, P P. Edwards, and P. A. Anderson, Chem. Commun., 2439 (2006).
(top)
Biomineralization of calcite in seashells
Organisms can control the, morphology, orientation, and size of crystalline blocks in biogenic crystals (e.g. shells) by means of organic molecules involved in the biomineralization process, though how they do this is unclear. One hypothesis is that the organic macromolecules adhere to specific planes and impede crystal growth in the perpendicular direction. Calcite prisms separated from two seashells were investigated on ID31 [3] and annealed for 30 min. at temperatures ranging between 50°C and 600°C. Geological calcite was used as a control. Structural parameters were extracted with high precision (about 10 ppm) by Rietveld refinement. These reveal anisotropic lattice distortions in biogenic samples compared to the geological counterpart.
Figure 2. Marked peak broadening on heating above 200 °C.
An intriguing result was the observation of a marked broadening of diffraction peaks of biogenic crystals annealed at temperatures above 200°C, figure 2, and a correlation of this broadening with a substantial relaxation of the anisotropic lattice distortions. Fitting a Voigt function provided estimates of the size of coherently scattered crystal blocks and averaged microstrain fluctuations. The broadening indicates a sharp drop in crystallite size and an increase in strain, which is interpreted as arising from the degradation of the structure-directing organic molecules. Changes in the microstructure correlate with the anisotropic lattice distortions, from which is was postulated that anisotropy of crystallite size and lattice distortions can be explained by assuming that during biomineralisation, organic macromolecules enter the calcite lattice oriented parallel to the (012) crystallographic plane. At the same time, the carboxylate groups of glutamic and aspartic residues replace those of calcium carbonate, producing maximum strain along the [001] direction. Understanding of the molecular mechanisms of such processes is a key issue towards fabrication of advanced materials via a biomimetic approach.
[3] The Microstructure of Biogenic Calcite: A View by High-Resolution Synchrotron Powder Diffraction. B. Pokroy, A. N. Fitch, and E. Zolotoyabko, Adv. Mater., 18, 2363 (2006).
(top)
References:
[1] A Mechanism for Non-stoichiometry in the Lithium Amide/Lithium Imide Hydrogen Storage Reaction. W. I. F. David, M. O. Jones, D. H. Gregory, C. M. Jewell, S. R. Johnson, A. Walton, and P. P. Edwards, J. Am. Chem. Soc., 129, 1594 (2007).
[2] Synthesis and crystal structure of Li4BH4(NH2)3. P.A. Chater, W. I. F. David, S. R. Johnson, P P. Edwards, and P. A. Anderson, Chem. Commun., 2439 (2006).
[3] The Microstructure of Biogenic Calcite: A View by High-Resolution Synchrotron Powder Diffraction. B. Pokroy, A. N. Fitch, and E. Zolotoyabko, Adv. Mater., 18, 2363 (2006).



