- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2013
- Soft condensed matter
- Thin film self-assembly behaviour of organic electronic molecule
Thin film self-assembly behaviour of organic electronic molecule
Conducting organic materials have promising potential applications in low-cost, solution-processable, flexible electronic devices such as photovoltaics and field-effect transistors. The efficiency of such devices is intimately related to the arrangement of the constituent molecules. The active layer of conducting polymer is typically required to be a few hundred nanometres thick. Within this “thin film” regime, the self-assembly behaviour can differ significantly from the bulk material due to additional interactions at the substrate-film and air-film interfaces. It is therefore of great importance to characterise and control the self-assembly of such molecules when confined to thin films.
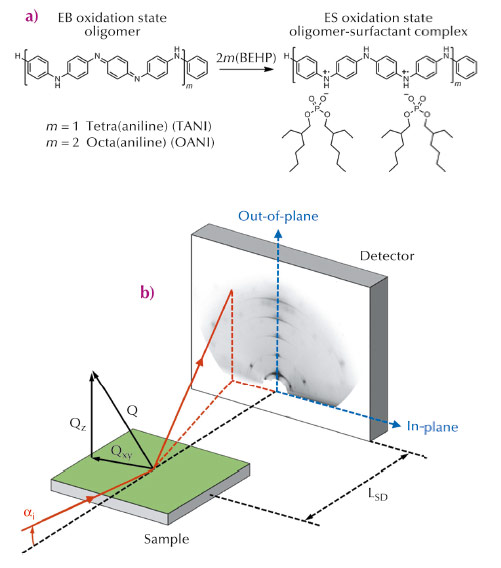
Our strategy for preparing well-ordered thin films is two fold. First, we use short oligomers of the conducting polymer poly(aniline) (PANI). Oligomers have the advantage of being more soluble (and thus processable) than PANI. Additionally, the oligomers can be prepared in high purity, overcoming the structural defects inherent in PANI [1]. Secondly, when combined with acid surfactants, oligo(aniline)s self-assemble into well-defined structures stabilised by secondary intermolecular interactions [2]. In this work, we have studied how oligo(aniline)-surfactant complexes self-assemble when confined to films of approximately 350 nm thick. The structure of these films has been examined at beamline BM28, using grazing-incidence X-ray scattering (GIXS) (Figure 80b). This technique allows us to elucidate the packing and orientation of molecules in extremely thin films.
 |
|
Fig. 80: (a) chemical structures of TANI (m = 1) and OANI (m = 2) and their complexes with the acid surfactant BEHP. (b) Grazing-incidence X-ray scattering geometry. |
Two oligomers were studied, TANI (four monomers) and OANI (eight monomers) (Figure 80a). Films of these oligomers and their complexes with the surfactant BEHP (bis(ethyl hexyl)phosphate) were prepared using a simple drop-casting method onto silicon substrates. In addition to studying the as-cast films, the structural response of the oligomer-surfactant films to thermal annealing was monitored in situ.
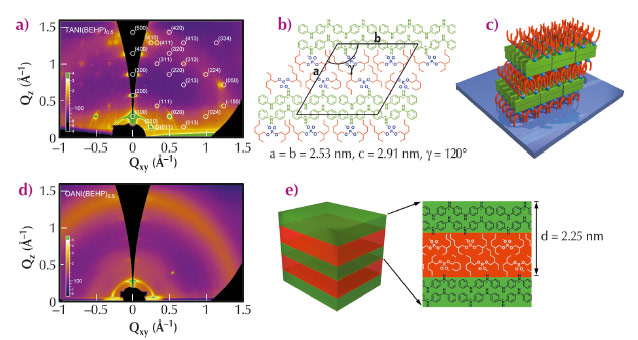
Films of the pure oligomers were completely disordered. When treated with BEHP, however, clear diffraction features were observed in the GIXS patterns, indicating the presence of well-ordered self-assembled structures in the thin film (Figure 81a and d). The GIXS pattern for TANI(BEHP)0.5 (TANI:BEHP molar ratio 1:2) presented spot-like diffraction features, indicating the formation of a highly ordered structure, with a high degree of orientation relative to the underlying substrate. The large number of reflections observed allowed us to determine that the molecules adopted a large unit cell, which was hexagonal in two dimensions (lattice parameters a = b = 2.53 nm, c = 2.91 nm, γ = 120°). It was assumed that the molecules adopted a bilayer arrangement in which bilayers of TANI molecules were separated by interdigitated alkyl tails from the BEHP molecules as depicted in Figure 81b. The high degree of preferential molecular orientation (Figure 81c) indicates that this system is likely to offer efficient charge-transport properties.
In the case of OANI(BEHP)0.5, the diffraction features in the GIXS patterns were broad rings rather than the spots seen for TANI(BEHP)0.5 (Figure 81d). This observation indicates that the ordered domains were randomly oriented on the surface. Furthermore, OANI(BEHP)0.5 adopted a simple lamellar structure, showing only one-dimensional order in contrast to the 3D order present for the TANI(BEHP)0.5 system as shown pictorially in Figure 81e. The difference in behaviour between these two systems arises from their differing molecular architectures. By doubling the chain length, the OANI oligomer has more degrees of freedom for rotation about phenyl-amine-phenyl bonds than TANI, which provides a greater entropic barrier to the formation of ordered thin films.
 |
|
Fig. 81: GIXS pattern (a), 2D lattice structure (b) and 3D surface orientation (c) for TANI(BEHP)0.5 thin film; GIXS pattern (d) and lamellar structure (e) for OANI(BEHP)0.5 thin film. |
Thermal annealing of the two films revealed further differences in their behaviour. At approximately 90°C the TANI(BEHP)0.5 film underwent an order–disorder transition. Upon cooling, the hexagonal structure reformed, however, the crystalline domains had an even greater preferential alignment relative to the silicon substrate. In the case of OANI(BEHP)0.5, the thermal energy was not great enough to induce any significant structural changes.
The detailed structural information of both films reveals, for the first time, how such oligomer-surfactant complexes self-assemble when confined to thin films at surfaces. The BEHP surfactant was pivotal in inducing structure formation and the resulting structures differed from those previously seen for the bulk phase materials. The exact morphology of the oligo(aniline) films could be altered through variation of the oligomer molecular architecture and thermal annealing. These results will guide the selection and processing of materials for future device fabrication.
Principal publication and authors
T.G. Dane (a), P.T. Cresswell (a), G.A. Pilkington (a), S. Lilliu (b), J.E. Macdonald (c), S.W. Prescott (a), O. Bikondoa (d, e), C.F.J. Faul (a) and W.H. Briscoe (a), Soft Matter 9, 10501-10511 (2013).
(a) School of Chemistry, University of Bristol (UK)
(b) Nano-Optics and Optoelectronics Research Laboratory, Masdar Institute of Science and Technology, Abu Dhabi (UAE)
(c) School of Physics and Astronomy, Cardi University (UK)
(d) XMaS, The UK-CRG Beamline at the ESRF (France)
(e) Department of Physics, University of Warwick (UK)
References
[1] Z.X. Wei and C.F.J. Faul, Macromol. Rapid Commun. 29, 280-292 (2008).
[2] T.G. Dane, P.T. Cresswell, O. Bikondoa, G.E. Newby, T. Arnold, C.F.J. Faul and W.H. Briscoe, Soft Matter 8, 2824-2832 (2012).



