- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Complex systems and biomedical sciences
- 3D imaging of whole cells using cryocooled coherent X-ray diffraction
3D imaging of whole cells using cryocooled coherent X-ray diffraction
X-ray imaging of whole cells using coherent diffraction could bridge the gap between optical and electron microscopy, but is limited by radiation damage. Cryocooling could be used to mitigate this limitation. Cryocooling is demonstrated here in conjunction with coherent diffractive imaging (cryo-CDI) to reveal the 3D structure of a parasitic cell with an overall spatial resolution better than 100 nm.
A long way from Robert Hooke’s first description of cells in cork, we are now experiencing a revolution in bioimaging. Optical microscopes are peering closer into cells than ever before, and electron microscopy approaches atomic resolution for single protein molecules. A type of X-ray microscopy known as coherent X-ray diffraction imaging (CDI) [1], which can recover a quantitative map of the 3D structure of a cell, promises to bridge these two imaging modalities.
3D CDI is similar to computed tomography (CT). It involves a synthesis of multiple projections of the same object from different angles. However, whereas CT relies on transmission images of an object, CDI interprets diffraction patterns from an object illuminated by a coherent X-ray beam (Figure 79). Because of this difference, CDI lacks phases, half of the data required to produce an interpretable image of the object. CDI uses computers as an X-ray lens to recover those phases and the structure of the object.
 |
|
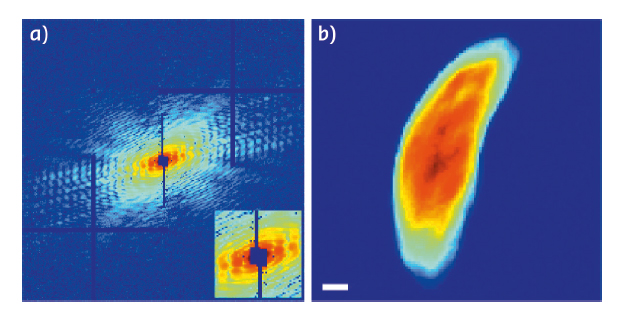
Fig. 79: A representative diffraction pattern taken at the 0° tilt angle (left panel) in which the horizontal and vertical bars are caused by the tiling of the 2 x 2 modules of the MAXIPIX detector. The inset shows an enlarged central part of the diffraction pattern (low spatial frequency region) where the missing data at the centre is due to a beam stop. A 2D projection (right panel) of the reconstructed 3D cell at the 0° tilt angle. Scale bar: 500 nm. |
CDI was first demonstrated experimentally in 1999 [2]. Since then, CDI has been used to image a range of samples including whole cells. Biological specimens pose a particular challenge for CDI since they scatter X-rays weakly and cannot tolerate the higher X-ray doses required to compensate for such weak scattering.
Cryogenic preservation techniques can safeguard a cell against irradiation, keeping it in a deeply frozen state during long-term exposure to X-rays. In a proof-of-principle experiment, we combined cryogenic techniques and 3D CDI at beamline ID10 to reveal the architecture of a parasite known as Neospora caninum. N. caninum primarily infects dogs, but is closely related to Plasmodium and Toxoplasma, the causative agents of malaria and toxoplasmosis in humans. The distinctive shape of N. caninum and the organisation of its inner structures are important for infection of its hosts and are highlighted in the recovered structure (Figure 80).
 |
|
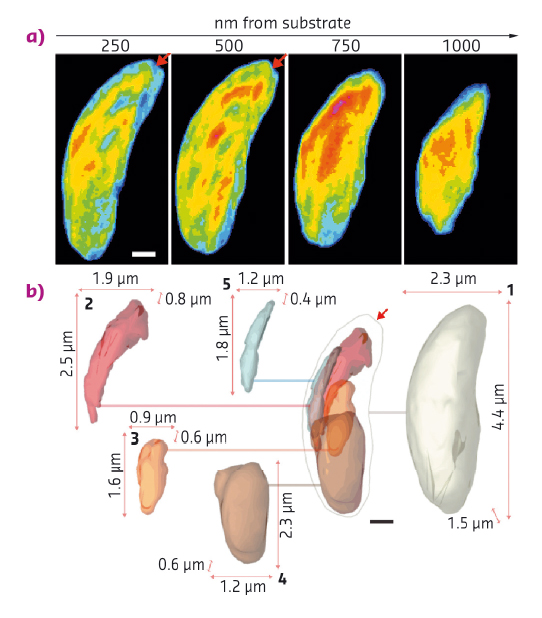
Fig. 80: A series of thin slices through the cell (top panel) at a distance ranging from 250 to 1000 nm away from the silicon nitride substrate, in which the red arrows indicate a conoid-like region. Images are false coloured according to normalised arbitrary density, lowest in blue, highest in red. A deconstructed isosurface model (bottom panel) shows features of the N. caninum cell as 2D projections of 3D renderings. At the centre, the boundary of the cell is shown, as well as other important organelles in their appropriate locations within the cell. A line extends from each of these regions to their cutouts, which are numbered and bound by the three lengths of the box that circumscribes them: 1. Cell boundary, 2. Rhoptries, 3. Apicoplast, 4. Nucleus, 5. Mitochondrion. Scale bars: 500 nm. |
This first experimental demonstration of cryo-CDI of frozen-hydrated cells using hard (8 keV) X-rays forecasts the routine and quantitative 3D imaging of whole cells with spatial resolutions in the tens of nanometres.
Principal publication and authors
Three-dimensional coherent X-ray diffractive imaging of whole frozen-hydrated cells, J.A. Rodriguez (a), R. Xu (b), C.-C. Chen (c), Z. Huang (d), H. Jiang (e), A.L. Chen (f), K.S. Raines (g), A. Pryor Jr (b), D. Nam (h), L. Wiegart (i), C. Song (h), A. Madsen (j), Y. Chushkin (k), F. Zontone (k), P.J. Bradley (f) and J. Miao (b), IUCrJ 2, 575–583 (2015); doi: 10.1107/S205225251501235X.
(a) UCLA-DOE Institute for Genomics and Proteomics, University of California, Los Angeles (USA)
(b) Department of Physics and Astronomy and California NanoSystems Institute, University of California (USA)
(c) National Sun Yat-sen University, Kaohsiung (Taiwan)
(d) Carl ZEISS X-ray Microscopy Inc., Pleasanton (USA)
(e) Shandong University, Jinan (People’s Republic of China)
(f) Department of Microbiology, Immunology, and Molecular Genetics, University of California, Los Angeles (USA)
(g) Department of Applied Physics, Stanford University (USA)
(h) Department of Physics, Pohang University of Science and Technology (South Korea)
(i) NSLS-II Photon Sciences Division, Brookhaven National Laboratory, Upton (USA)
(j) European X-ray Free Electron Laser, Hamburg (Germany)
(k) ESRF
References
[1] J. Miao et al., Science 348, 530–535 (2015).
[2] J. Miao et al., Nature 400, 342–344 (1999).



