- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Complex systems and biomedical sciences
- Imaging the microvascular network and the neuronal system in a mouse spinal cord
Imaging the microvascular network and the neuronal system in a mouse spinal cord
The repair capacity of an injured spinal cord relies on the interplay between the vascular network and the neuronal system. Our knowledge of how these two intricate systems overlap at the submicrometric level has been limited by the imaging tools available. Simultaneous imaging of both the vascular and neuronal architecture of the mouse spinal cord in 3D at scales spanning from millimetres to hundreds of nanometres using X-ray phase-contrast tomography has provided new insight into the neuro-vascular coupling. These results are expected to have a major impact on the investigation of neurodegenerative diseases.
Trauma and disease in spinal cord blood vessels and neurons lead to a wide range of neurodegenerative conditions. However, current imaging tools do not generate sufficient resolution and contrast to investigate these conditions or to understand the relationship between the vascular and neuronal systems [1]. Using a phase contrast imaging technique at beamline ID17, we have been able to visualise the complete architecture of both the vascular network and the neuronal system of the mouse spinal cord, imaging single elements of the networks such as microcapillaries and micrometric nerve fibres, axon-bundles and neuron soma. This imaging was carried out without sectioning or other destructive sample preparation procedures.
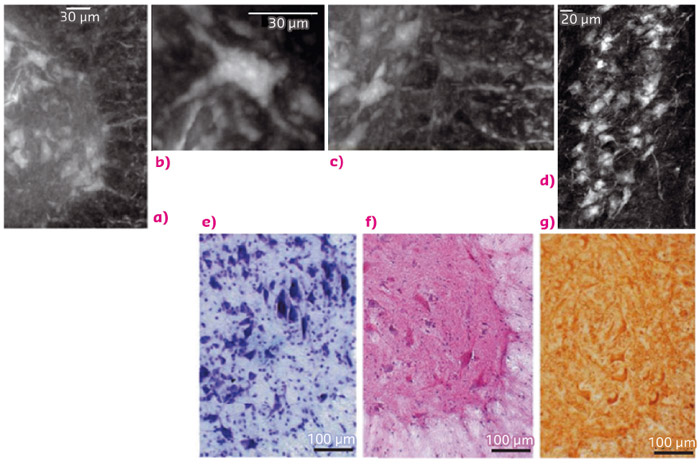
The combination of high spatial resolution (0.6 micrometre pixel size) with a large field of view for the tomographic images (1.2 mm in thickness) allows access to information on the large-scale architecture of the vascular network down to the level of the microcapillary organisation (Figure 91). This image also displays the long range connectivity of the neuronal system together with details down to the scale of a single neuron soma. We achieved a striking contrast between the white and the grey matter. Single neuron cells and nerve fibres are easily distinguishable at the white/grey matter interface in the anterior horn of the spinal cord. This spinal cord region is highly enriched with somatic motoneurons with a characteristic stellate morphology (Figure 91).
The single nerve fibre (Figure 91a, b, c) is imaged at the interface with the grey matter, while a longitudinal projection of the sample (about half a millimetre in length) at the same interface shows the connections between the cells, compatible with the dendrite branches (Figure 91d). Histological sections are shown for comparison in Figure 91e, f, g.
 |
|
Fig. 91: Neural population: (a) White/grey matter interface of a thick slab selected in the anterior horn of the lumbar-sacral spinal cord. (b) Longitudinal view (length: 0.5 mm) of the sample at the same interface. (c) Magnification of a single neuronal cell. (d) Image of one nerve fibre at the interface with the grey matter. For comparison, microscope histological images: (e) Histology (Nissl staining), (f) Histology (He/eo staining) and (g) Immunohistochemical analysis of SMI-32, a marker of motor neurons. |
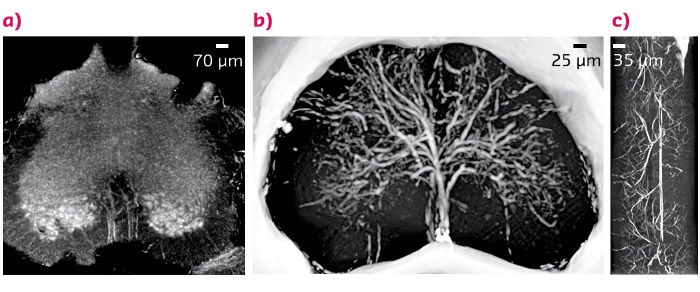
A twin sample was prepared with Microfil®, a contrast agent able to enhance the contrast of the microvasculature. The architecture of the vascular network was processed and is shown in Figure 92. The spatial distribution of the vascular network confirms that there is a small peripheral and large central vascular supply that is characteristic of the lumbar-sacral region.
 |
|
Fig. 92: (a) X-ray phase contrast microtomography of the lumbar-sacral region of the spinal cord (size: 1.2 mm), (b) Reconstructed volume of the lumbar-sacral region aft er administration of the Microfil contrast agent. The image was obtained with a pixel size of 3.5 μm. (c) Longitudinal view of (b), 1 mm long. |
Thus, our approach shows the potential of synchrotron X-ray phase contrast microtomography for pre-clinical investigations of the vascular system and degenerative pathologies of the central nervous system.
Principal publication and authors
M. Fratini (a,b), I. Bukreeva (c), G. Campi (d), F. Brun (e,f), G. Tromba (f), P. Modregger (g), D. Bucci (h), G. Battaglia (h), R. Spanò (i), M. Mastrogiacomo (i), H. Requardt (j), F. Giove (a,k), A. Bravin (j) and A. Cedola (c), Simultaneous submicrometric 3D imaging of the micro-vascular network and the neuronal system in a mouse spinal cord, Scientific Reports 5, 8514 (2015); doi: 10.1038/srep08514.
(a) “Enrico Fermi” Centre MARBILab c/o Fondazione Santa Lucia, Rome (Italy)
(b) Department of Science, Roma Tre University (Italy)
(c) Institute for Physical and Chemical Process-CNR c/o Physics Department at Sapienza University, Rome (Italy)
(d) Institute of Crystallography-CNR, Monterotondo, Rome (Italy)
(e) Department of Engineering and Architecture, University of Trieste (Italy)
(f) Elettra – Sincrotrone Trieste (Italy)
(g) Swiss Light Source, Paul Scherrer Institut, Villigen, and Centre d’Imagerie BioMedicale, Ecole Polytechnique Federale de Lausanne (Switzerland)
(h) I.R.C.C.S. Neuromed, Pozzilli (Italy)
(i) Department of Experimental Medicine, University of Genova and AUO San Martino - IST Istituto Nazionale per la Ricerca sul Cancro, Genova (Italy)
(j) ESRF
(k) Department of Physics, Sapienza University, Rome (Italy)
References
[1] M. Fratini et al., Journal of Tissue Engineering and Regenerative Medicine 8, 192 (2014).



