- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structural biology
- Structure and flexibility of endosomal Vps34 complex II
Structure and flexibility of endosomal Vps34 complex II
The phosphatidylinsositol 3-kinase complexes have fundamental roles in endocytic sorting, cytokinesis and autophagy. The structure of the endosomal Vps34 complex II was solved. This is a heterotetrameric assembly composed of Vps34, Vps15, Vps30/Atg6 and Vps38. In addition, hydrogen-deuterium exchange mass spectrometry provided insights into the way the complex adapts to membranes.
Phosphoinositide 3-kinases (PI3Ks) are a diverse family of enzymes that produce lipid second messengers in signalling pathways, with an ancient role in nutrient sensing. The PtdIns3P produced by the class III PI3K, Vps34, is critical for cargo sorting and membrane trafficking in endocytosis, phagocytosis, autophagy and cell division. This enzyme is present in several complexes. Complex I functions in autophagy and has four subunits: lipid kinase Vps34, protein kinase Vps15 (p150), Vps30/Atg6 (Beclin 1) and Atg14 (ATG14L). Complex II has Vps38 (UVRAG) instead of Atg14 and is involved in endocytic trafficking, as well as autophagy and cytokinesis in mammalian cells. A third complex containing Vps34 and Vps15 but not Vps30, Vps38 or Atg14 has been described, however, it might also include other previously uncharacterised proteins. The Vps34 complexes have been implicated in cancer, neurodegenerative diseases and ageing, which has led to a great interest in the development of specific inhibitors and activators of the complexes as well as in gaining a deeper understanding of how Vps34 complexes are regulated. To undepin these efforts, we have determined a crystal structure of Vps34 complex II and studied its membrane interactions by hydrogen-deuterium exchange mass spectrometry (HDX-MS).
Crystallisation required the presence of a nanobody specifically recognising Vps34. A final resolution of 4.4 Å was achieved at beamline ID29. To enable structure solution, high quality phase information was obtained using a Ta6Br12 derivative. High redundancy datasets were collected using the optimised data collection parameters from RADDOSE [1] together with the use of a mini-kappa goniometer to maximise the anomalous signal.
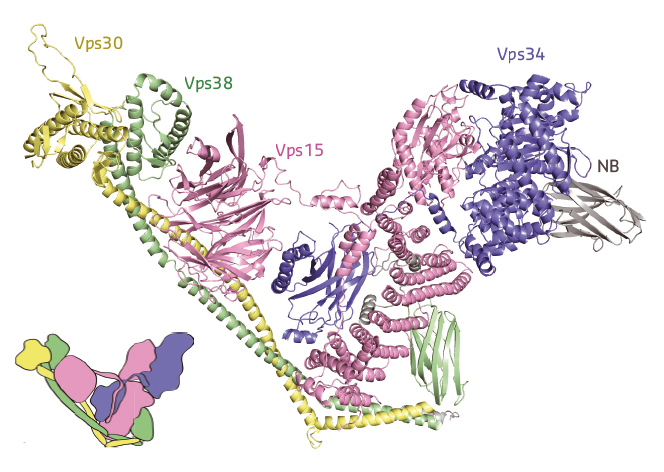
The structure revealed a Y-shaped complex, with the Vps34 C2 domain at its centre, engaging all the other subunits (Figure 115). In one arm, the lipid kinase Vps34 and the protein kinase Vps15 are intertwined, with their kinase domains directly interacting. Several catalytically important elements of Vps34, including the activation loop known to bind lipids, are found in close proximity to Vps15, which might indicate the way Vps15 modulates the kinase activity of Vps34. In the other arm, Vps30 and Vps38 form an extended bracket-like structure that fits on the Vps34/Vps15 catalytic heterodimer, suggesting a path for complex assembly.
 |
|
Fig. 115: X-ray crystallography shows that complex II has a Y-shape, with Vps30/Vps38 in one arm bracketing the Vps15/Vps34 heterodimer in the other arm. |
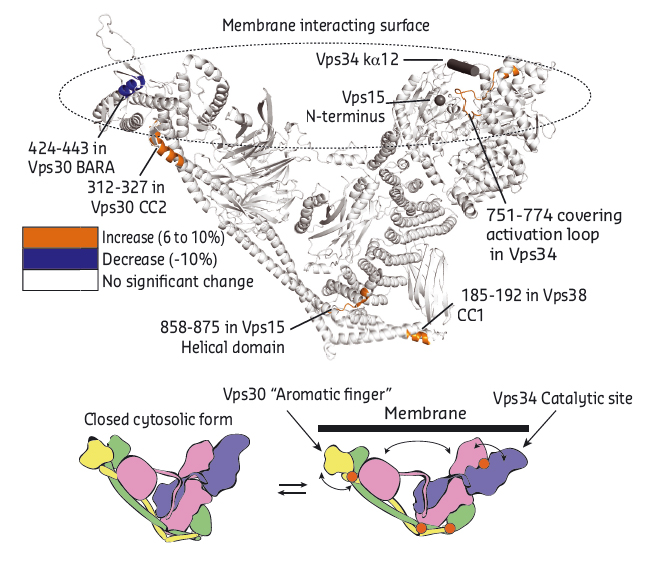
To understand how the complex functions on membranes, we performed HDX-MS analysis in the presence and the absence of lipids. We identified a loop in the C-terminal domain of Vps30 (referred to as “aromatic finger”) to be important for direct interaction with membranes (Figure 116). We have also observed several localised changes in HDX-MS rate away from the membrane interacting surface suggesting conformational changes in the complex upon membrane binding. These changes would be compatible with global opening/closing motions we observed in normal mode analysis.
 |
|
Fig. 116: Dynamics of Vps34 complex II on membranes. HDX-MS suggest conformational changes on membrane binding. |
Our kinase assays showed that complex I and II had very similar activities on small 100 nM vesicles which were significantly higher than the activity of Vps34/Vps15 heterodimer. An “aromatic finger” mutant reduced the activity of the complex II to that of Vps34/Vps15, highlighting the importance of both arms for membrane binding. On giant unilamellar vesicles (10 μm on average), only complex II was active, and its activity was abolished by the mutation of the “aromatic finger”.
Together, the structure of complex II, HDX-MS and kinase activity data allowed us to propose a model of how Vps34 complexes would adapt to membranes. The tips of the two arms would engage in membrane interaction, with the “aromatic finger” in one arm being required for the efficient catalysis by the other arm. In cells, complex I is involved in the initiation of autophagy where its activity would be restricted to small, highly curved tubular/vesicular structures, which could be related to the lack of activity on giant vesicles, exhibiting low curvature. On the other hand, complex II is involved in endocytic trafficking were it could encounter flat or curved membranes. To adapt to these differences in curvature, the two arms of the complex would open, providing the optimal interaction with the lipid substrate.
Principal publication and authors
Structure and flexibility of the endosomal Vps34 complex reveals the basis of its function on membranes, K. Rostislavleva (a), N. Soler (a), Y. Ohashi (a), L. Zhang (a), E. Pardon (b), J.E. Burke (c), G.R. Masson (a), C. Johnson (a), J. Steyaert (b), N.T. Ktistakis (d) and R.L. Williams (a), Science 350, 6257 (2015); doi: 10.1126/science.aac7365.
(a) MRC Laboratory of Molecular Biology, Cambridge (UK)
(b) Structural Biology Research Center, VIB, Brussels (Belgium)
Structural Biology Brussels, Vrije Universiteit Brussel (Belgium)
(c) Department of Biochemistry and Microbiology, University of Victoria (Canada)
(d) The Babraham Institute, Cambridge (UK)
References
[1] K.S. Paithankar et al., Acta Crystallogr. D 66, 381–388 (2010).



