- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structural biology
- Injector-based room-temperature serial crystallography of a membrane protein using synchrotron radiation
Injector-based room-temperature serial crystallography of a membrane protein using synchrotron radiation
To demonstrate how serial crystallography, developed in femtosecond timescales at X-ray free electron laser sources, can be implemented on a millisecond timescale at a synchrotron radiation source, membrane protein microcrystals in lipidic cubic phase have been injected into a X-ray microbeam. The resulting dataset allowed the structure to be solved at 2.4 Å resolution.
Serial femtosecond crystallography (SFX) as pursued at X-ray free electron laser (XFEL) sources relies mainly on injectors delivering the sample, such as protein microcrystals, in a fluid medium to pulsed X-rays. Simultaneously, vast datasets are recorded by fast area detectors specially designed for XFEL applications. A subset of these raw datasets contains the diffraction signal of the macromolecular sample (hits). Usually only a part of these hits can be matched to a single crystal lattice. These indexed frames are then merged to datasets suitable for macromolecular crystallographic structure solution methods.
 |
|
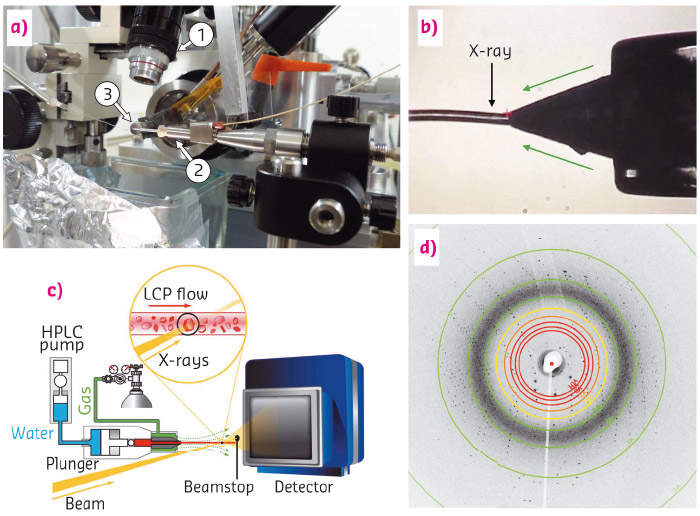
Fig. 131: (a) Experimental LCP-injector setup at ID13 showing: 1) the observation microscope, 2) the injector, 3) with its nozzle; (b) In situ microscope view of the LCP-jet during data collection; (c) schematic view of sample delivery and diffraction geometry; (d) diffraction pattern of bacteriorhodopsin with visible diffraction spots at up to 2.2 Å resolution. |
The aim of this study was to adapt the SFX method for use with microfocused synchrotron radiation at the microbranch of beamline ID13 (see Figure 131). Crystals of the membrane protein bacteriorhodopsin, grown in lipidic cubic phase (LCP) to be delivered by a LCP-injector [1], were chosen as a test case for realistic applications. LCP is a highly viscous medium which allows the jet to be tuned for very low stream velocities. The LCP jet of 50 µm in diameter was aligned horizontally, perpendicular to an X-ray beam with a spot size of a few micrometres. The bacteriorhodopsin microcrystals with sizes ranging from 5 to 40 µm traversed the beam at a speed of around 100 µm/s while serial diffraction data were recorded using a fast CCD area-detector at a frame rate of more than 10 Hz and exposure times of 10 or 25 ms. Often, the same microcrystal was observed in several consecutive frames. However, most of the time, each of those frames was recorded in a slightly different crystal orientation and therefore contributed to the diversity of the data set.
Data reduction was carried out in a very similar way to SFX methods using the programs Cheetah [2] and CrystFEL [3]. Starting from 13000 hits (~ 1% hit rate) containing bacteriorhodopsin diffraction patterns, about 5700 frames could be indexed, producing a data set which was subsequently used for structure solution to a resolution of 2.4 Å. The result is very similar to crystal structures obtained in previous studies based on standard cryogenic data collection. Although this experiment was carried out at room temperature, the refined structure showed only very minor signs of radiation damage. Furthermore, less than a milligram of protein was used for the entire data collection. These results suggest that synchrotron based serial crystallography with fast and sensitive state-of-the-art detectors operating in the kHz range could well develop into a powerful method for macromolecular crystal structure determination that is complementary regarding timescales (i.e. femtoseconds vs. milliseconds) with SFX applications. Moreover, large-scale synchrotron upgrade programmes such as ESRF-EBS should provide sources delivering a photon flux enhanced by several orders of magnitude when compared to current installations. These improvements will further enable serial microsecond crystallography, offering interesting opportunities for room-temperature kinetics studies, e.g. via laser excitation.
Principal publication and authors
Lipidic cubic phase serial millisecond crystallography using synchrotron radiation, P. Nogly (a), D. James (b), D. Wang (b), T.A. White (c) N. Zatsepin (c) A. Shilova (d), G. Nelson (b), H. Liu (b), L. Johansson (e), M. Heymann (c), K. Jaeger (a), M. Metz (c,f), C. Wickstrand (g), W. Wu (a), P. Bath (g), P. Berntsen (g), D. Oberthuer (c,f), V. Panneels (a), V. Cherezov (e), H. Chapman (c,h), G. Schertler (a,i), R. Neutze (g), J. Spence (b), I. Moraes (j,k,l), M. Burghammer (d,m), J. Standfuss (a) and U.Weierstall (b), IUCrJ 2, 168-176 (2015); doi: 10.1107/S2052252514026487.
(a) Laboratory for Biomolecular Research, Paul Scherrer Institute (Switzerland)
(b) Department of Physics, Arizona State University (USA)
(c) Center for Free-Electron Laser Science, DESY (Germany)
(d) ESRF
(e) Department of Integrative Structural and Computational Biology, Scripps Research Institute (USA)
(f) Centre for Ultrafast Imaging, Hamburg (Germany)
(g) Department of Chemistry and Molecular Biology, University of Gothenburg (Sweden)
(h) Department of Physics, University of Hamburg (Germany)
(i) Deparment of Biology, ETH Zürich (Switzerland)
(j) Membrane Protein Laboratory, Diamond Light Source (UK)
(k) Department of Life Sciences, Imperial College London (UK)
(l) Research Complex at Harwell Rutherford, Appleton Laboratory (UK)
(m) Department of Analytical Chemistry, Ghent University (Belgium)
References
[1] U. Weierstall et al., Nat. Commun. 5, 3309 (2014).
[2] A. Barty et al., J. Appl. Cryst. 47, 1118–1131 (2014).
[3] T.A. White et al., J. Appl. Cryst. 45, 335–341 (2012).



