- Home
- Users & Science
- Scientific Documentation
- ESRF Highlights
- ESRF Highlights 2015
- Structural biology
- RNA virus replication into ribonucleoproteins and its regulation elucidated from bunyavirus polymerase structure
RNA virus replication into ribonucleoproteins and its regulation elucidated from bunyavirus polymerase structure
The structural characterisation of the La Crosse bunyavirus polymerase by X-ray crystallography and cryo-electron microscopy provides a detailed description of the polymerase’s specific recognition of viral RNAs, the viral RNA genome’s allosteric regulation of polymerase activity and RNA traffic inside the polymerase during RNA synthesis. These discoveries explain many biochemical observations accumulated over decades of bunyavirus research, providing a detailed mechanistic understanding of bunyavirus RNA synthesis.
In 1943, a new virus was isolated from Aedes mosquitoes during an investigation of yellow fever in the Semliki forest, Uganda. This virus, called Bunyamwera, was the first of a new family of segmented negative stranded viruses that took the name Bunyaviridae. Since then, more than 350 strains have been isolated around the globe. Among them, La Crosse virus (LACV) is a mosquito-transmitted human pathogen that causes 50–100 cases of encephalitis per year in the USA, mainly affecting children [1].
Bunyavirus, in common with Arenavirus (e.g. Lassa virus) and Orthomixovirus (e.g. nfluenza virus), have several genomic RNA segments coated by viral nucleoproteins that are assembled with their polymerase to form circular RNA structures called ribonucleoproteins (RNPs), functional units of replication and transcription [2]. Transcription is carried out by the polymerases through a unique mechanism known as “cap snatching” [3] These functional similarities contrast with the sequence divergence between these viral polymerases. For example, the influenza virus polymerase is a heterotrimer [4] instead of a large monomeric polymerase (L protein) utilised by Arenavirus and Bunyavirus.
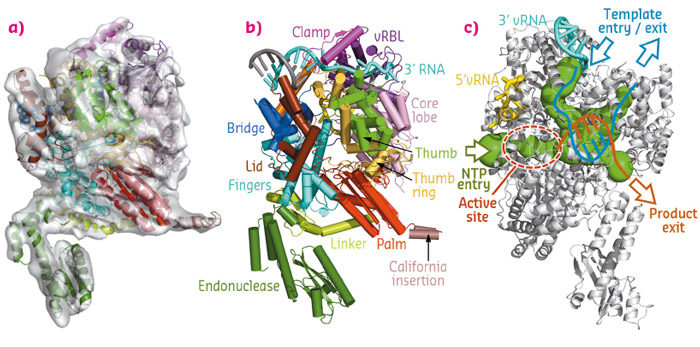
The structure of the LACV polymerase was obtained by protein crystallography using data collected at beamlines ID23-1 and ID29, and cryo-electron microscopy (Figure 133a, b) revealing several domains extending from the universally conserved RdRpol core, sharing a striking structural similarity with the influenza polymerase [4].
 |
|
Fig. 133: (a) 3D reconstruction of the LACV polymerase apo-L1750 by cryo-EM at 8.3 Å resolution. (b) Crystal structure of L1750 in complex with the 3’ (cyan) and 5’ (yellow) vRNA. (c) RNA traffic within the LACV polymerase cavities (in green) where the template (blue) and product (orange) enter and exit as single strands. |
The crystal structure of the L protein in complex with the viral RNAs clearly shows how the 3’ and 5’ RNA ends are bound at separate binding sites, and illustrates how the circularity of the RNP assembly is achieved (Figures 133b and 134a). The high specificity for the self-viral RNA recognition is mediated by extensive sequence specific protein-RNA contacts in positively charged clefts outside the catalytic site and close to the template entrance channel. The binding of the 5’-vRNA end has an allosteric effect on the active site that activates the polymerase by ordering the important ‘fingertips’ structural element in an active conformation.
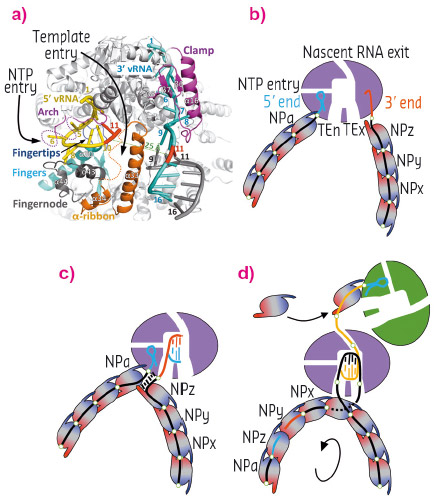
The internal cavity distribution allows for complete modelling of the RNA traffic inside the polymerase. This distribution implies that the template RNA to be read enters the polymerase and generates a double helix that after a few nucleotides is separated, the nascent RNA exits the polymerase near the cap-snatching machinery locus and the template RNA exits the polymerase proximal to the entry channel. The template RNA can then be reincorporated into the RNP with minimal disruption to the macro assembly structure (Figure 133c). This illustrates how the bunyavirus polymerase has adapted to work in the context of circular RNPs, maintaining their viral RNA as a single strand after duplication. Similar cavity distributions is found in the polymerase structures of influenza virus and vesicular stomatitis virus, therefore the conclusions revealed by the LACV polymerase structure can be extended to all NSV polymerases that work in an RNP context. We can thus propose a structure-based model of replication that might be extended to all sNSV and other related viruses such as VSV, Rabies or Ebola that also replicate in the RNP context [5] (Figure 134).
 |
|
Fig.134: (a) Representation of the LACV polymerase (grey) showing the disposition of key structural elements (labelled). The 5’ and 3’ vRNA ends are coloured in yellow and cyan, nucleotide 11 in each case is coloured in red to highlight their wide separation (>20 Å). (b) Schematic representation of panel (a), figure into the RNP. (c) With the 5’ end bound to the allosteric site for activation of RNA synthesis, a nascent cRNA begins to be synthesised. (d) Once the nascent c50 end emerges from the exit channel it can recruit an incoming apo-polymerase for encapsulating the progeny with incoming apo-NPs. |
Principal publication and authors
Structural insights into bunyavirus replication and its regulation by the vRNA promoter, P. Gerlach (a,b), H. Malet (a,b), S. Cusack (a,b) and J. Reguera (a,b), Cell 161, 1267–1279 (2015); doi: 10.1016/j.cell.2015.05.006.
(a) European Molecular Biology Laboratory, Grenoble Outstation (France)
(b) Unit of Virus Host-Cell Interactions (UMI 3265), University Grenoble Alpes-EMBL-CNRS (France)
References
[1] R.M. Elliott, Nat Rev Microbiol 12, 673-685 (2014).
[2] J. Reguera et al., Curr Opin Virol 5, 7-15 (2014).
[3] J. Reguera et al., PLoS Pathog 6, e1001101 (2010).
[4] A. Pflug et al., Nature 516, 355-360 (2014).
[5] J. Reguera et al., PNAS 110, 7246-7251 (2013).



